Safety Data Sheets (SDS): A Complete Guide to the 16 Sections, GHS, and Compliance
Learn what Safety Data Sheets (SDS) are, what the 16 GHS sections cover, and how to manage SDS compliance across your lab or research organization.

Download Whitepaper
Table of Contents
Table of Contents
TL;DR
A Safety Data Sheet (SDS) is a standardized 16-section document, required under OSHA’s Hazard Communication Standard and the UN’s Globally Harmonized System, that communicates chemical hazards, safe handling procedures, and emergency response information to anyone who works with hazardous substances.
- GHS standardization: The Globally Harmonized System of Classification and Labeling of Chemicals (GHS) is a UN-led framework that ensures chemicals are evaluated using consistent criteria worldwide, producing standardized labels, hazard pictograms, and Safety Data Sheets that eliminate confusion across borders and regulatory jurisdictions.
- The 16 SDS sections: Every compliant SDS follows a mandatory 16-section structure covering identification, hazard classification, composition, first-aid and firefighting measures, handling and storage guidance, exposure controls, physical and chemical properties, toxicological and ecological data, disposal, transport, and regulatory information.
- OSHA HCS 2024 update: OSHA’s revised Hazard Communication Standard aligns U.S. requirements with GHS Revision 7, with compliance deadlines in May 2026 for substance classifications and November 2026 for employer training and workplace labeling updates.
- Review and update cycles: SDS documents should be reviewed every 3 to 5 years and updated within 90 days when new hazard information emerges. Outdated sheets expose organizations to compliance violations, audit failures, and unsafe handling practices.
- Centralized SDS management: Electronic SDS management systems, integrated with chemical inventory tracking, ensure employees always access current versions, simplify audit preparation, and support faster emergency response compared to binder-based or folder-based approaches.
Introduction
A Safety Data Sheet (SDS) is a standardized 16-section document that provides comprehensive information about a chemical substance or mixture, including its hazards, safe handling and storage procedures, exposure controls, emergency response measures, and regulatory status. Required under OSHA's Hazard Communication Standard (29 CFR 1910.1200) and structured according to the United Nations' Globally Harmonized System of Classification and Labeling of Chemicals (GHS), Safety Data Sheets are the primary way chemical manufacturers, importers, and employers communicate hazard information to workers.
For research laboratories, manufacturing facilities, and scientific organizations, SDS documents are far more than a regulatory checkbox. They are the foundation of a chemical safety program that protects people, ensures compliance, and supports informed decision-making at every level, from the bench scientist handling reagents to the EHS director preparing for an audit.
This guide covers what Safety Data Sheets contain, how the GHS framework standardizes chemical hazard communication worldwide, what OSHA's 2024 HCS update means for your organization's compliance timeline, and how to build a sustainable SDS management process.
What is the Globally Harmonized System (GHS) and why does it matter for SDS?
The Globally Harmonized System of Classification and Labeling of Chemicals (GHS) is a United Nations-led international framework that standardizes how chemical hazards are classified and communicated. Before GHS, a single chemical could be classified differently depending on the country, requiring separate labels, separate Safety Data Sheets, and separate training materials for each regulatory jurisdiction.
GHS solves this by establishing consistent criteria for evaluating chemical hazards, resulting in standardized labels (including signal words, hazard statements, and pictograms) and a uniform 16-section SDS format that is recognized internationally.
The GHS framework is maintained by a UN Sub-Committee that regularly revises the guidance to reflect current scientific understanding and best practices. The most recent significant revision, GHS Revision 7, forms the basis of OSHA's 2024 update to the Hazard Communication Standard.
For organizations operating across multiple countries or regulatory environments, GHS alignment simplifies compliance by providing a single classification framework. A chemical classified and documented under GHS in the EU follows the same structure as one documented in the United States or Canada, reducing duplication and the risk of inconsistent hazard communication across sites.
Key benefits of GHS standardization:
- Consistent hazard classification criteria regardless of country
- Standardized SDS format (16 sections) used worldwide
- Universal pictograms and signal words that communicate risk visually
- Simplified cross-border trade and regulatory compliance for multi-site organizations
What are the 16 sections of a Safety Data Sheet?

Every compliant SDS follows a mandatory 16-section format. OSHA requires Sections 1 through 8 to contain information most critical for immediate safety decisions, while Sections 9 through 16 provide technical, environmental, and regulatory detail.
| Section | Title | What it covers |
|---|---|---|
| 1 | Identification | Product name, intended uses, supplier contact details, emergency phone number, and any restrictions on use. |
| 2 | Hazard(s) identification | Hazard classification, GHS pictograms, signal words, and standardized hazard and precautionary statements. |
| 3 | Composition / ingredients | Chemical identity and concentration of ingredients, including hazardous components above threshold concentrations. |
| 4 | First-aid measures | Response instructions by exposure route (inhalation, skin, eyes, ingestion), symptoms, and whether immediate medical attention is needed. |
| 5 | Fire-fighting measures | Recommended extinguishing agents, combustion hazards, and protective equipment for firefighters. |
| 6 | Accidental release measures | Personal precautions, environmental protection, and cleanup procedures for spills or leaks. |
| 7 | Handling and storage | Safe handling practices and proper storage conditions to prevent degradation or incompatible reactions. |
| 8 | Exposure controls / PPE | Occupational exposure limits, engineering controls (ventilation), and required personal protective equipment. |
| 9 | Physical / chemical properties | Appearance, odor, pH, boiling point, flash point, vapor pressure, solubility, and other measurable characteristics. |
| 10 | Stability and reactivity | Chemical stability, conditions to avoid, incompatible materials, and potential hazardous decomposition products. |
| 11 | Toxicological information | Acute and chronic health effects (irritation, carcinogenicity, reproductive toxicity, organ damage) and dose-response data. |
| 12 | Ecological information | Environmental impact: aquatic/terrestrial toxicity, persistence, bioaccumulation potential, and soil mobility. |
| 13 | Disposal considerations | Waste treatment methods, container disposal guidance, and special handling for contaminated materials. |
| 14 | Transport information | UN number, proper shipping name, transport hazard class, packing group, and special transport precautions. |
| 15 | Regulatory information | Applicable safety, health, and environmental regulations specific to the chemical beyond OSHA requirements. |
| 16 | Other information | SDS revision date, version history, abbreviation definitions, and references for further reading. |
OSHA's 2024 Hazard Communication Standard update: what changed for SDS
In May 2024, OSHA finalized a major update to the Hazard Communication Standard (HCS) to align U.S. requirements with GHS Revision 7. This is the most significant change to the HCS since the 2012 update that first adopted the GHS framework, and it directly affects how Safety Data Sheets are prepared, maintained, and communicated in the workplace.
The revised standard introduces new hazard classes, updates classification criteria, refines labeling requirements (including provisions for small containers), and adds specificity to SDS content requirements. OSHA expects these changes to reduce chemical-related workplace injuries and illnesses by improving the quality and clarity of hazard information available to employees.
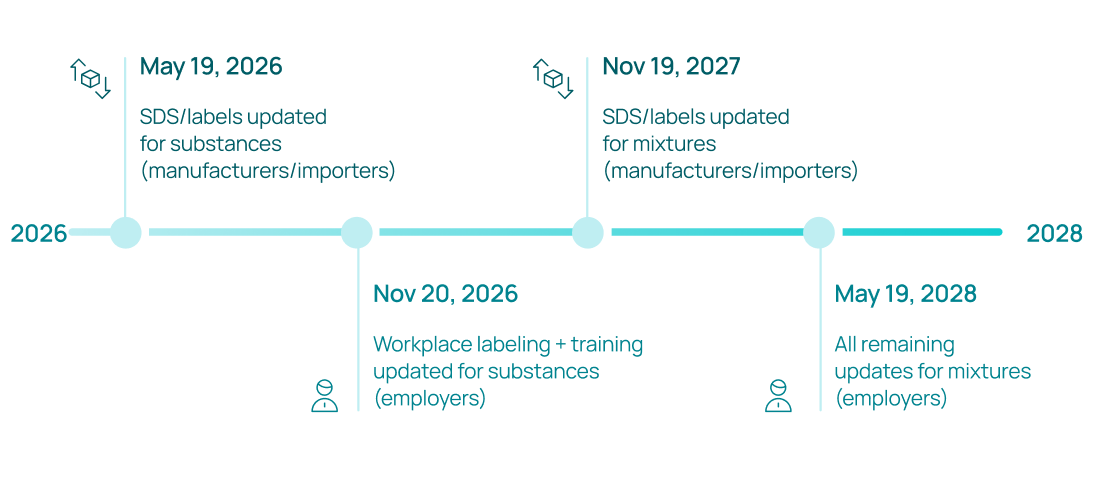
Key compliance deadlines (updated January 2026):
OSHA extended the original compliance dates by four months in January 2026 to give organizations additional time to implement the changes. The current deadlines are:
- May 19, 2026: Chemical manufacturers, importers, and distributors must update SDS documents and labels for pure substances to align with the revised HCS.
- November 20, 2026: Employers must update workplace labeling, employee training programs, and written hazard communication programs for substances.
- November 19, 2027: Manufacturers and importers must update SDS and labels for chemical mixtures.
- May 19, 2028: Employers must complete all remaining updates for mixtures.
During the transition period, organizations may comply with the 2012 HCS, the revised 2024 HCS, or a combination of both. However, mixed compliance requires careful coordination to ensure that labels, SDS documents, and training materials remain consistent.
For research institutions and multi-site organizations, these deadlines mean that SDS management processes, chemical inventory systems, and employee training programs all need to be reviewed and updated within the next 12 to 24 months. Organizations that rely on manual tracking methods, paper binders, or disconnected spreadsheets face a significantly higher risk of falling behind.
For organizations preparing for these compliance deadlines, building a structured approach to chemical inventory and SDS management becomes critical. This includes how chemicals are tracked, how SDS documents are maintained, and how updates are reflected across sites and teams.
Our webinar on building a scalable chemical inventory program walks through how to structure intake, reconciliation, and compliance reporting workflows that help keep SDS records consistent and up to date as requirements evolve:
Alright. Hello, everyone. We are still watching the participants climb here, but we'll get started. We only have thirty minutes and, really appreciate and value your time. So we're gonna kick things off here. So first, just very excited to welcome such a diverse group. We've got a lot of people joining, well over a hundred registrations and participants and actively climbing as we watch that number go up. And you're coming to us from all sizes of organizations, institutions across the globe. Really appreciate that. You took some time to join us today. My name is Jon Zibell, and I am the VP of strategic alliances here at SciSure. And I'm joined by Sarina Schwartz-Hinds, who is our principal product manager and our chemical inventory guru. She will be educating us on a number of things throughout the conversation. Sarina put this best as we were talking about this topic, she said, I want to give chemical inventory management the respect it deserves. That kind of hit me. Is not a simple thing. It's not an easy thing. It does take change management. It does take intention and deliberate practice to get this right and deliberate process to get it right. Our goal today is to arm you with some talking points so you can go back to your organizations and institutions and bring about some positive change. As I said, great inventory programs ensure compliance. They're just not a glorified spreadsheet that tracks materials, but they actually automate the compliance and get you there. So we're going to incorporate findings from well over our one thousand customers and try to translate those into a roadmap that you can use. I think the key for today is take a step forward. This isn't a boil the ocean kind of scenario here. We want you to be able to take a step forward. So as we have on the screen here, you know, when we think about chemical inventory programs, we're gonna touch on this, what we call the trifecta. It's critical to have a great process and program for intake, clear accurate records of what's coming in. You then need to take those records of what those materials are and attach them to a chemical profile using a database, a catalog. What do we know about this chemical? What hazards are related to this chemical? And the third part here is gonna be the compliance and regulatory reporting. Not only do how do we keep our people safe? I mean, this can come down to PPE and different training requirements, but also how do would where do these, chemicals belong in our regulatory reporting? How are we gonna get those over to our regulatory reports so that we stay compliant and we reduce risk? And we wanna think about all three of these right from the start. So let's let's dive into it. Right? John and I have been working with customers during chemical inventory for, like, ten to fifteen years here. We have a passion for chemical inventory. Doesn't mean everyone always loves to do it, but we wanna make it, right, scalable. It's easy to do. Let's walk through it. What what are what are the processes? What do you need to do regardless of how you do chemical inventory today? Whether that's spreadsheets, whether that's software, whether that's paper. Right? And where we start is capturing data accurately at the source. Connect with that tightly is understanding the safety and compliance implications of every chemical you're entering. Right? This isn't just like inventory at a store. Right? Though there's some overlap with that. These are materials that we need to produce regulatory reports on. So this is why we wanna think about, right, all of right. Think about the end game when we're starting inventory. And how do we do this in a way that brings efficiency? Right? We wanna be ready for these reports. We wanna keep that in mind as we're building our inventory up. So let's talk about, right, how do we do this first part? If we're not using software, right, typically, you know, what does this day to day look like? Right here, you know, we have our chemical. We're on our laptop computer. Probably have a spreadsheet. Right? A lot of folks start with Excel when they just have maybe, like, fifty to a few hundred bottles here. I'm typing in my chemical, the CAS number, probably it to I'm always googling those. Right? If I if I was doing this on a spreadsheet or maybe I'm typing it in from the bottle, just getting the basics. Right? We need that chemical name, that CAS number, physical state, the size of that bottle with the right units. This is where efficiency and accuracy and having a good process. This is where that can fall apart, right, actually immediately here. If we aren't, you know, if we aren't doing this in a way that is gonna work with us as our company or organization grows here. So this is feeling familiar to a lot of folks, a lot the customers that we work with right before they start using ChemTracker and SciSure here. A lot of them are using spreadsheets, or if it's not a spreadsheet, some sort of digital program where they're kinda doing the same thing, right, where where it has this. And we wanna have this decentralized. So in this next, you know, next column here, we're talking about where do we want this to go, and where's the industry evolving? Right? There's a ton of a ton of options out there for chemical inventory. And we've seen AI and automation. This is where this is where chemical inventory trends are going. Is something we've been really passionate about here at SciSure. We introduced ChemSnap dot ai sometime in the last year where instead of that spreadsheet, you can just take a photo. I mean, I think it makes inventory actually kind of fun. Definitely saves a lot of time. Right? Everyone on this call, right, whether in EHS, lab ops, whether you're a scientist, we know you have other things to do. Right? You have a lot on your mind. And if inventory is a big part of your job, there's probably a lot of chemicals you have to get through each day. So we wanna speed that process up. K. This next step, the chemical profile catalog. I'm so passionate about this. This this it adds efficiency, but also about from a safety standpoint, we need to know, right, what are the implications of each chemical? And these aren't obvious. Right? I have a chemistry background. You can't just hand me a bottle and have me automatically know, okay. You know, I know which EPA list that's on. I know that's flammable, all of this. So this is a little walk through about, right, if I'm doing this on my spreadsheet or, again, even a software program that doesn't have this built in. That's typically what I'm doing. Right? I'm googling that. Maybe I'm trying to put into chat GPT. Right? Gotta be kinda careful with that these days. I don't think the AI is quite there to put all this. Going to my safety data sheet. Right? This one from Sigma Aldridge, chemist myself. Right? Love Sigma Aldridge here. I'm trying to paste a link to the safety data sheet. And this is just kind of a pain. Right? It's error prone. And then I'm trying to copy and paste these hazards. Right? Looking at, right, of course, GHS data, NFPA data. Right? A lot of folks still use that. A lot of states require it on door signs, but it's not always on the newer safety data sheets. Trying to make sure I have that safety data sheet for OSHA compliance here, looking at physical hazards, health hazards, environmental hazards. Know this is a lot of work to to do by hand. Right? We're losing efficiency. We're probably losing accuracy. Right? Because this process might have you be kind of rushed. Or or you're spending a lot of money. Right? A lot of FGs on this. I wanna think ahead. Right? Our next step, how are you gonna do reports? And this part, right, again, you're doing these lookups over and over again. The industry is going towards dynamic catalogs. Right? This is ChemTracker from the very start has been built with this this core process in mind. Right? ChemTracker started at Stanford University, years ago here. It's been in SciSure for a long time, and we knew that this dynamic catalog, which you're gonna hear more about, is key. Right? So you don't have to do all those lookups so that we can get to our reports. Right? I'm sure everyone's been in the position they're trying to scramble on this. A lot of pressure. Right? A lot of risk. A lot on the line. What you're doing and, again, whether you use spreadsheets or whether you're using a program, bring together all these, right, all these inventories. You're like, okay. Where are my gaps? And in my diagram here, is nicely highlighted. We know in real life it's, I mean, it's never that simple. I have to figure it all out, then figure out who do I call, who do I email. You know, at a large company, you might be even hopping on an airplane. You're going across town. You're driving across the state, right, to to a different site here to track this data down. This is where, right, having gaps in your data is gonna break your process. And then when it comes time to do, right, our tier two or RTK reports, fire code reports, maximum allowable quantities, is an area where a lot of our customers come in because they get caught realizing they just don't right? They don't have that data. They're not ready. They don't have control areas. They don't know what is vulnerable one a. Right? Because we know this data. It's not one to one with GHS data on our safety data sheets. It's its own definitions in some cases. Or maybe, right, just an internal courier audit. Right? And this is where your EHS program gets that stress test. And this is often where you find, you know, we need this regulatory knowledge. We're tired of doing these manual lookups, and we need to have collaboration, right, across different departments, across, you know, across the organization here so we can scale and not have these these blind spots anymore. Right? You can't afford it. So this is, again, that stress test, and this is where we wanna see research and lab ops and EHS tightly connected because this this is critical in so many areas. And, Jon, this brings me to the trifecta here. Right? What are we looking at? Yeah. So I think, you know, I do wanna recap a little bit of what Sarina said there. You know, when you look at your program, look for those three foundational pillars, which is what we have in front of us right now. You know, with these, you're able to build that scalable program that reduces the administrative burden and all those errors Serena talked about. You know, think about how many times if you have all of your chemical inventory tools in quotes or sources in different areas, the manual transcribing of that data coming across or or someone typing it in to a free form page is where you see those errors. So, when we think about this, we're really back to this this sort of chem tracker trifecta mindset. If you have that good point of entry, it's efficient, it's not hard to do. If the scientists and researchers are like, This is a pain in the butt, not going to do it. Well, what? They won't do it. Going it. Yeah, good luck, thirty:thirty We all know that. So that's where you have to be able to provide them. If you show them you're investing in solutions that are built for them, built for the lab that are actually going to work, we find they actually use it. It's not that they don't want to use it, it's that we're giving them wrong solutions. You have to have a central source of truth across all of your spaces. We start with customers all the time and it's in five disparate tools. How can EHS lab ops and leadership possibly do their job? How can the risk management team do their job if the data's in five different places? Going on to the next part here, this is really where, to me, is the most powerful part of what our system does. You have to figure out how to get to this point. It's validating and getting that information about the chemical. It's great to say we have it, but now what? What are the implications of having this on-site? Is it going to put us over an MAQ? Are we in trouble with the fire code compliance? Those are all the questions that if you don't have this trifecta in order and you don't have it seamlessly tied together, however you go about doing that, you end up creating a bunch of headaches and problems down the road. Things as simple as auto assigning SDS. When a chemical comes through the door, if you have a great profile, you know which chemical it is, you know the hazards around it, you can get the SDS assigned to it right away. Yeah. And this is one hey, John, if you don't mind me jumping in. Yeah. Right? We note automation. That's for our our customers. Right? When you're using ChemTracker, it feels seamless. It feels like magic. This isn't something you can't automate all of these chemical profiles. We still have people doing some of this work. I think one day the AI will get there. Trust me. We'll definitely wanna use it. Right? It'll save us a a lot of resources there. But until that point, right, it's really important to us that this is a high quality, trustable data, not just, like, random data, not just, you know, pulled out. It's a trustworthy, right, validated source here, and I think that's gonna write power reports here. Yeah. And, you know, when you think about the reporting, so now you know everything about this chemical. You can actually start to automate where it belongs in your regulatory reporting, which reports does it belong on, what do we need to know about it, even things as simple as what volumes do we have across a single control area. All of those things become critical because those are the manual touch points. If that doesn't automate and someone misses that, you're obviously not putting your chemicals into the proper reports. And I think to summarize this is a lot of what Serena said. We look at manual touch points. Manual touch points in disparate systems create gaps, gaps create risk. One thing that we focus on all the time when we look at this from a product lens is how can we shine light on the areas that are a known gap or a known risk? Just shining light and knowing it's there doesn't mean we've actually fixed it. But now we can say, okay, now we know where the gaps are and where the issues are and where the problems are, and our team will get to work on figuring out how do we connect this together, how do we automate it so that you confidence in your compliance records and your reporting and you feel good that there is this seamless triangle without all that manual touch, which ends up creating the gaps. And to tie this all together, you know, we think of this trifecta. It's not just a trifecta of chemical inventory. It's actually a trifecta of the roles that it impacts. If the scientists don't want to use the tool at intake and they don't want to engage with it and it's not built for them and it's clunky or the UI is terrible, we hear that all the time. If someone says, hey, we're looking for a new solution. The scientists just can't use this. It's clunky. It's old. It's not working. They're not gonna use it. That's the reality. That has a a, you know, downstream impact on lab operations. They don't know what inventory is about to run out. Then they come back and say, lab ops, where's my chemicals? Lab managers, where's my chemicals? And they're not able to do their job effectively. That in turn, EHS doesn't know what's in the lab. It's not going into your regulatory compliance. And then somebody orders the wrong thing, And now you're over your MAQs and you're out of your fire code compliance, and it's not showing up on reports. So think about this in terms of you you wanna audit your program to figure out, do we have these three core components in place? However we get there, even if you take a step into it and it's not fully automated, get the three core components in place, then look at your roles throughout the lab and think about scientists, lab operations, and EHS and the impact it's having on them. Alright. So we know, right, this matters for everyone. We need the science to keep going. Right? Let's if you can't find what you're looking for, can't order what you want, running into issues here. Yeah. So, you know, thinking about this sort of, trifecta here of safety and compliance, we've actually been running a benchmark study across now thirty two. And I think this morning we actually had our thirty third one come through. So this is an ongoing benchmark we've been looking at. And the key here is this actually helps us from a product standpoint. How do we continue to drive down the time it takes someone to perform a task? How do we continue to make things more efficient? But also ensure that the data you know, when a customer looks at where they were day one, at the starting point before they turned on SciSure, you can do this with whatever solution you have, take an audit of your program. But what we did was we said, where are our customers day one and where are they at six months, one year, eighteen months? Not only how much time do we save them, but let's look at their compliance rates. How many, training requirements are met because we're tracking things more clearly? How often are are reports generated that are accurate and on time? So we did a deep benchmark study across a number of our customers, which we'll embed in the data here throughout the conversation. Yeah. Data is key here. Right? You can't measure. You can't change it. So let's talk about getting data in. Right? How do we get inventory data in? First, ChemSnap AI. I talked about this very briefly, right, with just a photo. It's kind of the stuff of dreams. Right? You take a picture of the bottle, the data inputs in there. You can also use barcodes and RFID tags. This is gonna help with that reconciliation. Right? Because inventory, we wish it was a onetime process. It's not. Right? You're always gonna have to be updating that data. Let's look through it. How do we add inventory in? This is with ChemTracker here. Add with camera ChemSnap dot ai. It's our most popular way to add inventory today. It'll auto populate a number of these fields from that label. Right? Name of that chemical connects it to our database. We're gonna see here. We're gonna have predefined drop downs about keeping our data clean, about who's owning this, where it's stored. And if I I am using barcodes or RFID tags, ChemTracker out of the box, you can use either or a combination. I see some of that, data scanned in here as well. Okay. Let's talk about data. Right? I just said you can't measure it. You can't change it. We saw with our customers, right, almost a seventy four percent reduction in the amount of time to add a container. Might be thinking, oh, this is just a few minutes, but, you know, this efficiency matters. Right? It's it's through one container that'll scale across, right, hundreds, thousands, right, hundreds of thousands. We see inventories in the two, three hundred thousand dollar three hundred thousand bottle range. I guess a lot higher in the in the dollar numbers there. And then that's about just adding inventory and reconciliation. I just say this number blew me away. Right, John? I'm sure for you too. Right? We see twenty hours to half an hour. So, like, ninety seven and a half percent. Crazy. Yeah. It's awesome to see that. I think, you know, one part of this is, hey. The technology can do great things, but the team that that's working in the technology, as Serena said earlier, you know, we're empowering people to be much more efficient, effective in their job to drive safety, to drive compliance. As she mentioned, know, when we look at the data, we're really trying to figure out that compounding effect. What's cool about this is you have a really this snowball effect or compound interest of the effort you put in upfront. Think about how many times across your institution or organization someone is adding a container to inventory. These stats say six point five minutes to one point seven minutes. Yeah, it's cool if it's a few times, but what we're talking about here is the scientist who's saying, I got to go perform this task. And every single time I do it multiple times a day, a week, a month, a year, I'm gaining back five minutes or so every single time I do that. You multiply that by the number of scientists you have across your organization, and that's really where the compound interest lines up. It's that power of a program that you put in place. Because today, even with ten scientists, if you're giving them a simple solution to use, they will be very grateful for that five minutes. When you compound it and you start to scale and grow, you have exponential ROI to the organization. And reconciliation rate twenty hours. This is, more than two day two two business days. Right? So how do we how do we get these numbers? Right? Accuracy of automation. These are also better records we're getting. Right? It's easy to use. We're gonna have this hazard data in place. Right? So we're gonna see more time saving even once that inventory is in because when you add inventory, it'll connect to our validated chemical profiles here. So let's talk about these chemical profiles. Right? This is this is the key. When you enter data or when we import your data, right, from spreadsheet, we're gonna tie to our chemical property database. This is available only in ChemTracker. It's you know, we have about two million safety data sheets with auto matching to safety data sheets. We provide hazards, storage groups for chemical compatibility, density data. That's gonna be key for fire codes, CAS numbers, and synonyms. Right? So any name on that label, wanna make sure we can look it up. Right? One person enters dichloromethane, another person enters methylene chloride. Now you can search by either name and find that. We'll do unit conversions and then bring in all this regulatory data for you. So this, again, it all comes in from the chemicals. Right? Here we are at this example I was just talking about, we can type in even that abbreviation DCM. This is one, right, again, for researchers. This is also key for each one of these profiles. This is an example for dichloromethane. It's definitely one of our hot chemicals on the mind after that EPA rule. Right? Common solvent turned a high hazard, high abbreviation, high supervision here. We have structure data. You could also add SOPs with safety data sheets. Physical properties. Right? Densey mentioned this is key so that you can do those unit conversions automatically. Storage handling for EPA tier two reports. And this is then just regulations just for this chemical. Alright? This is state, local, DOT. I won't say we have every single regulation in the United States ever, but we have a lot of them. Right? There's a lot here. And, again, these are the ones for just one chemical. Highlighting here right now is the ones for fire code reporting. We know those. Right? Not always one to one with GHS. We have that available for our customers in the database. And all of this comes together, right, to build in, these validated profiles. Hey, Serena. I did wanna pause for a second. We have a a question in the chat that I think is worth clarifying. Someone asked, hey. If I if I use the photo and I take a photo of the chemical container, will it actually bring the chemical profile over to the container and assign the SDS to it? You're saying I can do that with a photo. That's the beauty of the trifecta of what we built here. You know, just to tell a story to really drive this home, this idea came about. We were at a site visit with someone who was about to deploy, ChemTracker. They had not yet turned it on. And while we were walking through on the prep meeting, we had a scientist take a box of chemicals over to the bench and start pulling them out and hitting their keyboard. The head of safety said, let's go take a look at what's going on here. Turned out she was searching the chemical profile like, hey, what hazards am I getting exposed to here? Walking through a list and jotting it down on a note on her desktop so she knew what she was working with. And, you know, the Chemsnap AI AI idea came out of those conversations where we said we have to do better. We cannot have our scientists spending any time on this. We want them to come in, take a picture of the container, hit the database, grab the SDS, tell them everything they need to know, and then one step further, put that into the health and safety and regulatory compliance reports. So something as simple as once that's in there, I just wanna search for my peroxide formers. You know? That's the beauty of this feature. So that's called ChemSnap AI, and that's something that's available now. Basically kicks off everything that we see here. Yeah. So this this is what makes it scalable. Right? You know, we've seen inventory everywhere. Right? Your lab is not a store. It's not generic inventory. You need to understand those chemicals, and that is what ChemTracker does here, whether that's with photo, whether that's with spreadsheet, whether, you know, maybe you're typing a catalog number if you you don't have a mobile device here. So we have all that together right here. Alright. If we're running again, right, how do we get these numbers? It's from, right, again, the the multiple steps here, that trifecta. So we have ChemSnap. We have other ways to get the data and bar codes and RFID options to make reconciliation easier, tie to those validated chemical profiles with SDS auto match, and then you can pull these, you know, these real time reports and queries. Again, correcting chemical data, looking at over two work days a month. Right? Think about what else you can do with that. Right? Do with that time here. So when we think about real time reporting, right, this is the end game. This is what we're trying to get to. Ninety nine point four percent time savings that we've seen, and this is, again, with a real customer study here going from over twenty four hours to seven point two minutes to to pull report. This stat actually didn't surprise me when I'd seen this, right, collected in. Think about, you know, just even someone asking, okay. You know, what are all our peroxide formers? What do we have that's, you know, like a certain level of toxicity and then doing those unit conversions? I've seen firsthand for customers, right, out how hard this is if they don't, you know, I don't have a program that can do this like ChemTracker does. We can do tier two. We can do RTK, including threshold calculations. For our code report, you'll have your control areas. You can see percentages of MAQs here. Right? Because we have this data in here, and you can do it by whatever location set you need to here. In terms of that real, you know, real time reporting here, I have over nine hundred regulations, these automated hazard calculations with the MAQ ones. Right? Again, fire code is is often everyone's brain, especially in the Boston area here. A lot of ways you can get to this type of data, whether it's, you know, export to spreadsheet or API. And what we're showing here, this is exactly what it would be like for you. Right? Once we have your data into ChemTracker, if you're not a customer already, select any regulation in our database and convert into exact units you need. Labs have everything in metric units. You can run-in pounds, gallons, and cubic feet here and get the data that you need out. Yeah. So, Serena, we can go to the next slide. This is a refresh on the on the trifecta and the triangle here. We're just gonna walk through a little bit of, you know, in the detail of what we covered as a recap. But when you think about ChemTracker, you know, we built it for this trifecta. The whole point is that if we can't create this seamless experience from the efficient intake to that validated chemical profile that you can trust. You know, this is an area we spend a lot of time and a lot of resources at SciSure investing in that chemical profile database, so that our customers don't have to think about it. It's an out of sight, out of mind. You license SciSure and ChemTracker, not something you have to think about. And then moving into real time reporting, did see there's so much in the chat. A question about, we report by laboratory? Can we report by building? Absolutely. You'll start to see the data broken out in different groups so that you can query it. You can find out, search the data. I want to look for pyrophoric, so I want my peroxide formers. I want to know where they are. I want to know who's working with them. All those things that you can query the data. And on top of that, you can push all of it into your regulatory reporting. So we talked earlier about our goal is to get rid of those blind spots and those gaps. You have to first start by shining a light on what it is, and that's probably the exercise I'm sure most of you will go back and do. Hey. What do we have today, and how do we get ourselves to a point that we can meet this sort of trifecta, process here? And, really, it's not just efficiency and speed. It's the accuracy and the productivity that we're after. This is what we do. We understand chemicals. So I think Mark's gonna be joining here to help us with the q and a. John, maybe you can give a little overview of this while we're pulling up our questions here. Yeah. So if you think about, we just wanna you know, many of you come to us from different parts, whether you're an existing customer or future customer. We have our research side of the house, which is your ELN, LIMS, sample management, all the solutions and tools to really help the scientists and researchers, advance the science, innovate, and do it safely and efficiently and have reproducibility in their daily work. And then we have the health and safety and lab operations side. We have a number of solutions. We also have marketplace and integrations. I think I saw some vendors joined us today and partners. If you'd like to integrate your solution into SciShore, we have an open marketplace and ecosystem where vendors can plug in. That gives our customers access to a number of different solutions to continue to build that seamless experience inside of SciShore. Yeah, Mark, if you don't mind me jumping in, saw this question from Jackie here that I really wanna start with. Jackie wrote, I also wonder since the chemical hazards are classified from using GHS from the SDS while the MAQ report follows IFC, does that mean there's a link mapping between GHS and IFC in the system? So for anyone not familiar, IFC is international fire code, basis of most, you know, most fire codes in the state states and cities in the United States. So our chemical database is different than others in that it's not data just blindly pulled from safety data sheets. I wish it was. Right? That'd be a lot easier to do. Fire code data in particular, most of the time, right, it's not one to one with the GHS classifications. This is for things like toxicity in particular. We have this is, I'd say, the hardest thing to train for our curators for the database. They're looking at LD 50s, LC 50s, calculating it into the right time range, things like that to get those fire code assignments, right? Because unfortunately they're not on the safety data sheets here. That's great. And also guys, there's another question. It's regarding MAQ again from Erica. She's mentioning for MAQ calculations, does this software allow for assigning laboratories a percentage of the MAQ threshold? Yeah. There's a way you could set this up. You actually have control over what the MAQs are for a given control area or control zone. So in that case, probably the way that we'd set up the location so the labs could be limited just a certain section of the room and basically have their own their own MAQ percentage there. Mark, I wanna grab a real quick one here. I see that. Yep. Contractors working on-site utilize ChemTracker, or can we give them access IDs and passcodes? Absolutely. We work with almost any vendor that you have on-site now, we work with. We always say we play nice in the sandbox. If their job is to get your program compliant to be safe, to be efficient, we are the tech stack that helps. So actually many of the sites you might, go to or visit, it's often our software powering the effort going on there. And then we work with contractors, vendors, consultants to make sure that they have best in class technology to do their job. K. And I see a few a few questions about SDS updates. I see one from Mindy asking if it will notify stakeholders and chemical owners if there's an SDS update. One from Jackie about the SDS and ChemTrackers database updates automatically when a new version is released. So what we have every right? We know SDS. Right? This isn't a onetime job. So each each chemical, each bottle, right, we recheck to see if there's a newer SDS or a better match at least once a year. They'll automatically update that within the system. Not notification of that per se, but you'll be to see that, right, see that SDS and see the last date that that was was updated there. Someone also asked, I wanna answer this one about reconciliation. I would ask this. That's a great system. Thank you, I would. It's really kind. Said after I have my inventory done, can I multiscan the bottles for reconciliation? There's something you can do with barcodes or RFID tags. You know? Right? You might have, like, ten thousand bottles in one lab. So it's designed to designed to scale there. And I also see another important question that you might be able to touch base on, Sarina. The question is, can you import my data if I'm currently using another system? Yes. Absolutely. Say, it's pretty rare that, you know, a new customer comes in and they don't have anything for inventory on occasion, but usually folks at least have a spreadsheet. Most software systems out there can provide some sort of export to spreadsheet, and we can work with you to figure out, right, how that fits in our template. We don't have quite as many required fields as some systems do to, you know, to import. So typically, right, even if you don't have CAS numbers, we can import your data, and we can match to the database so you can get those safety data sheets. You can get that hazard data and those reports right away here. Yeah. Sarina, I just wanna go add to that answer. So one thing that's really important is during your discussion with our team, we'll do really a discovery call and work with you to diagnose what problems are you facing, what's existing today, how do we improve it, how do we set up metrics so that we can track the output, not just stand up the system, but then as we shared earlier, monitor our progress. One thing that we will do is we always assign a dedicated implementation team to every single customer. So that team will help you with getting any data into the system, setting it up the right way, checking back with you, making sure that you have this highly successful implementation and deployment. And in the past ten years, we have one hundred percent success rate in deployment and industry best retention. That's not a coincidence. It's because of the effort we put in to understand what you need from us to build a program that meets those needs and then to come back and implement it and deliver it the way that you wanted us to deliver it to your success criteria. So if there's any questions around that, I'm happy to set up a call. We do not want to lose customers, and we take that to heart. Some companies will say, Hey, we're going get ten through the door, we'll lose a couple as we go. We do not want to lose a single customer. So we put a ton of effort into that implementation, making sure we get it right. If you get it right out of the gate and you're successful, it works in perpetuity. If you don't get it right out of the gate and it's a tough rollout, it's not going to work. And that's why our model is to be very white gloved and hand holding out of the gate to make sure that we get to success quickly. I see another question here I want to touch on. Rui Feng, I apologize if I don't have the pronunciation of your name right here, but could you please introduce the function of tracking time sensitive chemicals if you have? Yes. So among the lists we have in ChemTracker, we have one that's time sensitive that might cover things, you know, that with, you know, with age could degrade in a way that could be hazardous, as well as a separate list of peroxide formers. So absolutely something you can get to here as well as tracking expiration dates, seeing what's expired, all of that here. Right. Yeah. I mean, we still have a full house here. We still have a lot of questions, but I think in in the interest of time, Mark, maybe we'll wrap up here. We'll coordinate, some follow-up on the remaining questions here. Thank you so much everyone taking the time Thank you all. Really appreciate it. Have a great day.

Who needs Safety Data Sheets and when are they required?
Under OSHA's Hazard Communication Standard, SDS requirements apply across the chemical supply chain:
Chemical manufacturers and importers must prepare an SDS for every hazardous chemical they produce or bring into the country and provide it to downstream customers with the first shipment.
Distributors must ensure the correct, current SDS accompanies every shipment of hazardous chemicals to downstream buyers.
Employers must obtain and maintain a current SDS for every hazardous chemical present in the workplace. These must be readily accessible to employees during every work shift, not stored in a locked cabinet or buried in a shared drive folder that nobody can find during an emergency.
This requirement applies broadly. It covers research laboratories, manufacturing facilities, hospitals, agricultural operations, maintenance shops, and any workplace where employees handle chemicals beyond typical consumer use. OSHA citations for Hazard Communication violations are consistently among the agency's most frequently issued, and penalties for serious violations can reach $16,550 per instance.
For scientific organizations managing hundreds or thousands of chemical containers across multiple labs, buildings, or campuses, maintaining current SDS documentation is a significant operational challenge that manual processes alone cannot reliably support.
How to manage Safety Data Sheets effectively across your organization
Having SDS documents on file is only part of the requirement. The real compliance challenge is ensuring they are current, accessible, and connected to your chemical inventory. Here is what an effective SDS management process looks like in practice.
The difference between paper-based and digital SDS management isn't just convenience. It affects compliance readiness, emergency response time, and whether your team can actually find the right information when it matters.
- Keep SDS documents linked to your chemical inventory. An SDS is only useful when it can be quickly located for the specific chemical in question. The most reliable approach is an electronic system that automatically links SDS records to individual chemical containers, searchable by chemical name, CAS number, or manufacturer. This eliminates the common problem of having an SDS binder that doesn't match what's actually on the shelf.
- Review and update on a defined cycle. The GHS recommends reviewing SDS information every 3 to 5 years, even when no new hazard data has been reported. EU and OSHA regulations also require updates within 90 days of learning significant new information about a chemical's hazards. Without a systematic review process, outdated sheets accumulate and create silent compliance gaps.
- Ensure accessibility meets regulatory expectations. OSHA requires SDS to be readily accessible during every work shift. "Readily accessible" means employees can locate the relevant SDS quickly, without needing to track down a supervisor, search through folders, or navigate a confusing directory. Electronic SDS databases, particularly those integrated with chemical inventory management, meet this standard by allowing search-based access from any workstation, lab terminal, or connected device.
- Connect SDS to training and onboarding. Every employee who works with or near hazardous chemicals must be trained on how to read, understand, and access Safety Data Sheets. This training should be documented and refreshed whenever new chemicals are introduced or when SDS content changes due to reclassification. A training management system that tracks completion and triggers reminders simplifies this for organizations with large or distributed teams.
- Prepare for audits proactively. Regulators expect to see an up-to-date chemical inventory with matching SDS for every hazardous chemical on-site. They also check that employees know where to find SDS documents and how to read them. Organizations that manage SDS digitally, with integrated compliance reporting, can demonstrate audit readiness in minutes rather than scrambling to assemble paper trails.
For a deeper look at what regulators typically ask during chemical safety inspections, see 5 Common Questions From Chemical Regulators (and How to Address Them).
Organizations building or upgrading their chemical inventory program can also explore SciSure's Chemical Inventory Management Playbook for a step-by-step framework covering intake, reconciliation, SDS management, and regulatory reporting.
GHS hazard pictograms: visual communication of chemical risks

The GHS uses a set of nine standardized pictograms, each enclosed in a red diamond-shaped border, to visually communicate specific categories of chemical hazards. These pictograms appear on both chemical labels and in Section 2 of every SDS.
Each pictogram represents a distinct hazard type: health hazards (such as carcinogenicity or respiratory sensitization), physical hazards (such as flammability or explosiveness), and environmental hazards (such as aquatic toxicity). The visual standardization means that a worker in any country can recognize the hazard category at a glance, regardless of language.
For lab managers and safety and compliance officers, understanding pictograms is essential for proper chemical storage (ensuring incompatible materials are separated), for selecting appropriate PPE, and for making informed decisions about which chemicals can be used in shared spaces.
A reference guide to all GHS pictograms is available from OSHA's Quick Card on Pictograms.
Conclusion
Safety Data Sheets are one of the most important documents in any chemical safety program. They standardize how hazard information is communicated, protect the people who work with chemicals every day, and form the basis of regulatory compliance under OSHA's Hazard Communication Standard.
With OSHA's revised HCS aligning U.S. standards to GHS Revision 7 and compliance deadlines approaching in 2026 and 2027, now is the time for organizations to review their SDS management processes. That means verifying that every chemical on-site has a current SDS, ensuring employees can access and understand those documents, and connecting SDS records to the broader chemical inventory and safety infrastructure that supports day-to-day operations.
The organizations that treat SDS management as an ongoing operational process, rather than a one-time filing task, are the ones that stay audit-ready, respond faster in emergencies, and build a genuine culture of chemical safety.
Ready to see SciSure in action?
No commitment · Free consultation
Introduction
A Safety Data Sheet (SDS) is a standardized 16-section document that provides comprehensive information about a chemical substance or mixture, including its hazards, safe handling and storage procedures, exposure controls, emergency response measures, and regulatory status. Required under OSHA's Hazard Communication Standard (29 CFR 1910.1200) and structured according to the United Nations' Globally Harmonized System of Classification and Labeling of Chemicals (GHS), Safety Data Sheets are the primary way chemical manufacturers, importers, and employers communicate hazard information to workers.
For research laboratories, manufacturing facilities, and scientific organizations, SDS documents are far more than a regulatory checkbox. They are the foundation of a chemical safety program that protects people, ensures compliance, and supports informed decision-making at every level, from the bench scientist handling reagents to the EHS director preparing for an audit.
This guide covers what Safety Data Sheets contain, how the GHS framework standardizes chemical hazard communication worldwide, what OSHA's 2024 HCS update means for your organization's compliance timeline, and how to build a sustainable SDS management process.
What is the Globally Harmonized System (GHS) and why does it matter for SDS?
The Globally Harmonized System of Classification and Labeling of Chemicals (GHS) is a United Nations-led international framework that standardizes how chemical hazards are classified and communicated. Before GHS, a single chemical could be classified differently depending on the country, requiring separate labels, separate Safety Data Sheets, and separate training materials for each regulatory jurisdiction.
GHS solves this by establishing consistent criteria for evaluating chemical hazards, resulting in standardized labels (including signal words, hazard statements, and pictograms) and a uniform 16-section SDS format that is recognized internationally.
The GHS framework is maintained by a UN Sub-Committee that regularly revises the guidance to reflect current scientific understanding and best practices. The most recent significant revision, GHS Revision 7, forms the basis of OSHA's 2024 update to the Hazard Communication Standard.
For organizations operating across multiple countries or regulatory environments, GHS alignment simplifies compliance by providing a single classification framework. A chemical classified and documented under GHS in the EU follows the same structure as one documented in the United States or Canada, reducing duplication and the risk of inconsistent hazard communication across sites.
Key benefits of GHS standardization:
- Consistent hazard classification criteria regardless of country
- Standardized SDS format (16 sections) used worldwide
- Universal pictograms and signal words that communicate risk visually
- Simplified cross-border trade and regulatory compliance for multi-site organizations
What are the 16 sections of a Safety Data Sheet?

Every compliant SDS follows a mandatory 16-section format. OSHA requires Sections 1 through 8 to contain information most critical for immediate safety decisions, while Sections 9 through 16 provide technical, environmental, and regulatory detail.
| Section | Title | What it covers |
|---|---|---|
| 1 | Identification | Product name, intended uses, supplier contact details, emergency phone number, and any restrictions on use. |
| 2 | Hazard(s) identification | Hazard classification, GHS pictograms, signal words, and standardized hazard and precautionary statements. |
| 3 | Composition / ingredients | Chemical identity and concentration of ingredients, including hazardous components above threshold concentrations. |
| 4 | First-aid measures | Response instructions by exposure route (inhalation, skin, eyes, ingestion), symptoms, and whether immediate medical attention is needed. |
| 5 | Fire-fighting measures | Recommended extinguishing agents, combustion hazards, and protective equipment for firefighters. |
| 6 | Accidental release measures | Personal precautions, environmental protection, and cleanup procedures for spills or leaks. |
| 7 | Handling and storage | Safe handling practices and proper storage conditions to prevent degradation or incompatible reactions. |
| 8 | Exposure controls / PPE | Occupational exposure limits, engineering controls (ventilation), and required personal protective equipment. |
| 9 | Physical / chemical properties | Appearance, odor, pH, boiling point, flash point, vapor pressure, solubility, and other measurable characteristics. |
| 10 | Stability and reactivity | Chemical stability, conditions to avoid, incompatible materials, and potential hazardous decomposition products. |
| 11 | Toxicological information | Acute and chronic health effects (irritation, carcinogenicity, reproductive toxicity, organ damage) and dose-response data. |
| 12 | Ecological information | Environmental impact: aquatic/terrestrial toxicity, persistence, bioaccumulation potential, and soil mobility. |
| 13 | Disposal considerations | Waste treatment methods, container disposal guidance, and special handling for contaminated materials. |
| 14 | Transport information | UN number, proper shipping name, transport hazard class, packing group, and special transport precautions. |
| 15 | Regulatory information | Applicable safety, health, and environmental regulations specific to the chemical beyond OSHA requirements. |
| 16 | Other information | SDS revision date, version history, abbreviation definitions, and references for further reading. |
OSHA's 2024 Hazard Communication Standard update: what changed for SDS
In May 2024, OSHA finalized a major update to the Hazard Communication Standard (HCS) to align U.S. requirements with GHS Revision 7. This is the most significant change to the HCS since the 2012 update that first adopted the GHS framework, and it directly affects how Safety Data Sheets are prepared, maintained, and communicated in the workplace.
The revised standard introduces new hazard classes, updates classification criteria, refines labeling requirements (including provisions for small containers), and adds specificity to SDS content requirements. OSHA expects these changes to reduce chemical-related workplace injuries and illnesses by improving the quality and clarity of hazard information available to employees.

Key compliance deadlines (updated January 2026):
OSHA extended the original compliance dates by four months in January 2026 to give organizations additional time to implement the changes. The current deadlines are:
- May 19, 2026: Chemical manufacturers, importers, and distributors must update SDS documents and labels for pure substances to align with the revised HCS.
- November 20, 2026: Employers must update workplace labeling, employee training programs, and written hazard communication programs for substances.
- November 19, 2027: Manufacturers and importers must update SDS and labels for chemical mixtures.
- May 19, 2028: Employers must complete all remaining updates for mixtures.
During the transition period, organizations may comply with the 2012 HCS, the revised 2024 HCS, or a combination of both. However, mixed compliance requires careful coordination to ensure that labels, SDS documents, and training materials remain consistent.
For research institutions and multi-site organizations, these deadlines mean that SDS management processes, chemical inventory systems, and employee training programs all need to be reviewed and updated within the next 12 to 24 months. Organizations that rely on manual tracking methods, paper binders, or disconnected spreadsheets face a significantly higher risk of falling behind.
For organizations preparing for these compliance deadlines, building a structured approach to chemical inventory and SDS management becomes critical. This includes how chemicals are tracked, how SDS documents are maintained, and how updates are reflected across sites and teams.
Our webinar on building a scalable chemical inventory program walks through how to structure intake, reconciliation, and compliance reporting workflows that help keep SDS records consistent and up to date as requirements evolve:
Alright. Hello, everyone. We are still watching the participants climb here, but we'll get started. We only have thirty minutes and, really appreciate and value your time. So we're gonna kick things off here. So first, just very excited to welcome such a diverse group. We've got a lot of people joining, well over a hundred registrations and participants and actively climbing as we watch that number go up. And you're coming to us from all sizes of organizations, institutions across the globe. Really appreciate that. You took some time to join us today. My name is Jon Zibell, and I am the VP of strategic alliances here at SciSure. And I'm joined by Sarina Schwartz-Hinds, who is our principal product manager and our chemical inventory guru. She will be educating us on a number of things throughout the conversation. Sarina put this best as we were talking about this topic, she said, I want to give chemical inventory management the respect it deserves. That kind of hit me. Is not a simple thing. It's not an easy thing. It does take change management. It does take intention and deliberate practice to get this right and deliberate process to get it right. Our goal today is to arm you with some talking points so you can go back to your organizations and institutions and bring about some positive change. As I said, great inventory programs ensure compliance. They're just not a glorified spreadsheet that tracks materials, but they actually automate the compliance and get you there. So we're going to incorporate findings from well over our one thousand customers and try to translate those into a roadmap that you can use. I think the key for today is take a step forward. This isn't a boil the ocean kind of scenario here. We want you to be able to take a step forward. So as we have on the screen here, you know, when we think about chemical inventory programs, we're gonna touch on this, what we call the trifecta. It's critical to have a great process and program for intake, clear accurate records of what's coming in. You then need to take those records of what those materials are and attach them to a chemical profile using a database, a catalog. What do we know about this chemical? What hazards are related to this chemical? And the third part here is gonna be the compliance and regulatory reporting. Not only do how do we keep our people safe? I mean, this can come down to PPE and different training requirements, but also how do would where do these, chemicals belong in our regulatory reporting? How are we gonna get those over to our regulatory reports so that we stay compliant and we reduce risk? And we wanna think about all three of these right from the start. So let's let's dive into it. Right? John and I have been working with customers during chemical inventory for, like, ten to fifteen years here. We have a passion for chemical inventory. Doesn't mean everyone always loves to do it, but we wanna make it, right, scalable. It's easy to do. Let's walk through it. What what are what are the processes? What do you need to do regardless of how you do chemical inventory today? Whether that's spreadsheets, whether that's software, whether that's paper. Right? And where we start is capturing data accurately at the source. Connect with that tightly is understanding the safety and compliance implications of every chemical you're entering. Right? This isn't just like inventory at a store. Right? Though there's some overlap with that. These are materials that we need to produce regulatory reports on. So this is why we wanna think about, right, all of right. Think about the end game when we're starting inventory. And how do we do this in a way that brings efficiency? Right? We wanna be ready for these reports. We wanna keep that in mind as we're building our inventory up. So let's talk about, right, how do we do this first part? If we're not using software, right, typically, you know, what does this day to day look like? Right here, you know, we have our chemical. We're on our laptop computer. Probably have a spreadsheet. Right? A lot of folks start with Excel when they just have maybe, like, fifty to a few hundred bottles here. I'm typing in my chemical, the CAS number, probably it to I'm always googling those. Right? If I if I was doing this on a spreadsheet or maybe I'm typing it in from the bottle, just getting the basics. Right? We need that chemical name, that CAS number, physical state, the size of that bottle with the right units. This is where efficiency and accuracy and having a good process. This is where that can fall apart, right, actually immediately here. If we aren't, you know, if we aren't doing this in a way that is gonna work with us as our company or organization grows here. So this is feeling familiar to a lot of folks, a lot the customers that we work with right before they start using ChemTracker and SciSure here. A lot of them are using spreadsheets, or if it's not a spreadsheet, some sort of digital program where they're kinda doing the same thing, right, where where it has this. And we wanna have this decentralized. So in this next, you know, next column here, we're talking about where do we want this to go, and where's the industry evolving? Right? There's a ton of a ton of options out there for chemical inventory. And we've seen AI and automation. This is where this is where chemical inventory trends are going. Is something we've been really passionate about here at SciSure. We introduced ChemSnap dot ai sometime in the last year where instead of that spreadsheet, you can just take a photo. I mean, I think it makes inventory actually kind of fun. Definitely saves a lot of time. Right? Everyone on this call, right, whether in EHS, lab ops, whether you're a scientist, we know you have other things to do. Right? You have a lot on your mind. And if inventory is a big part of your job, there's probably a lot of chemicals you have to get through each day. So we wanna speed that process up. K. This next step, the chemical profile catalog. I'm so passionate about this. This this it adds efficiency, but also about from a safety standpoint, we need to know, right, what are the implications of each chemical? And these aren't obvious. Right? I have a chemistry background. You can't just hand me a bottle and have me automatically know, okay. You know, I know which EPA list that's on. I know that's flammable, all of this. So this is a little walk through about, right, if I'm doing this on my spreadsheet or, again, even a software program that doesn't have this built in. That's typically what I'm doing. Right? I'm googling that. Maybe I'm trying to put into chat GPT. Right? Gotta be kinda careful with that these days. I don't think the AI is quite there to put all this. Going to my safety data sheet. Right? This one from Sigma Aldridge, chemist myself. Right? Love Sigma Aldridge here. I'm trying to paste a link to the safety data sheet. And this is just kind of a pain. Right? It's error prone. And then I'm trying to copy and paste these hazards. Right? Looking at, right, of course, GHS data, NFPA data. Right? A lot of folks still use that. A lot of states require it on door signs, but it's not always on the newer safety data sheets. Trying to make sure I have that safety data sheet for OSHA compliance here, looking at physical hazards, health hazards, environmental hazards. Know this is a lot of work to to do by hand. Right? We're losing efficiency. We're probably losing accuracy. Right? Because this process might have you be kind of rushed. Or or you're spending a lot of money. Right? A lot of FGs on this. I wanna think ahead. Right? Our next step, how are you gonna do reports? And this part, right, again, you're doing these lookups over and over again. The industry is going towards dynamic catalogs. Right? This is ChemTracker from the very start has been built with this this core process in mind. Right? ChemTracker started at Stanford University, years ago here. It's been in SciSure for a long time, and we knew that this dynamic catalog, which you're gonna hear more about, is key. Right? So you don't have to do all those lookups so that we can get to our reports. Right? I'm sure everyone's been in the position they're trying to scramble on this. A lot of pressure. Right? A lot of risk. A lot on the line. What you're doing and, again, whether you use spreadsheets or whether you're using a program, bring together all these, right, all these inventories. You're like, okay. Where are my gaps? And in my diagram here, is nicely highlighted. We know in real life it's, I mean, it's never that simple. I have to figure it all out, then figure out who do I call, who do I email. You know, at a large company, you might be even hopping on an airplane. You're going across town. You're driving across the state, right, to to a different site here to track this data down. This is where, right, having gaps in your data is gonna break your process. And then when it comes time to do, right, our tier two or RTK reports, fire code reports, maximum allowable quantities, is an area where a lot of our customers come in because they get caught realizing they just don't right? They don't have that data. They're not ready. They don't have control areas. They don't know what is vulnerable one a. Right? Because we know this data. It's not one to one with GHS data on our safety data sheets. It's its own definitions in some cases. Or maybe, right, just an internal courier audit. Right? And this is where your EHS program gets that stress test. And this is often where you find, you know, we need this regulatory knowledge. We're tired of doing these manual lookups, and we need to have collaboration, right, across different departments, across, you know, across the organization here so we can scale and not have these these blind spots anymore. Right? You can't afford it. So this is, again, that stress test, and this is where we wanna see research and lab ops and EHS tightly connected because this this is critical in so many areas. And, Jon, this brings me to the trifecta here. Right? What are we looking at? Yeah. So I think, you know, I do wanna recap a little bit of what Sarina said there. You know, when you look at your program, look for those three foundational pillars, which is what we have in front of us right now. You know, with these, you're able to build that scalable program that reduces the administrative burden and all those errors Serena talked about. You know, think about how many times if you have all of your chemical inventory tools in quotes or sources in different areas, the manual transcribing of that data coming across or or someone typing it in to a free form page is where you see those errors. So, when we think about this, we're really back to this this sort of chem tracker trifecta mindset. If you have that good point of entry, it's efficient, it's not hard to do. If the scientists and researchers are like, This is a pain in the butt, not going to do it. Well, what? They won't do it. Going it. Yeah, good luck, thirty:thirty We all know that. So that's where you have to be able to provide them. If you show them you're investing in solutions that are built for them, built for the lab that are actually going to work, we find they actually use it. It's not that they don't want to use it, it's that we're giving them wrong solutions. You have to have a central source of truth across all of your spaces. We start with customers all the time and it's in five disparate tools. How can EHS lab ops and leadership possibly do their job? How can the risk management team do their job if the data's in five different places? Going on to the next part here, this is really where, to me, is the most powerful part of what our system does. You have to figure out how to get to this point. It's validating and getting that information about the chemical. It's great to say we have it, but now what? What are the implications of having this on-site? Is it going to put us over an MAQ? Are we in trouble with the fire code compliance? Those are all the questions that if you don't have this trifecta in order and you don't have it seamlessly tied together, however you go about doing that, you end up creating a bunch of headaches and problems down the road. Things as simple as auto assigning SDS. When a chemical comes through the door, if you have a great profile, you know which chemical it is, you know the hazards around it, you can get the SDS assigned to it right away. Yeah. And this is one hey, John, if you don't mind me jumping in. Yeah. Right? We note automation. That's for our our customers. Right? When you're using ChemTracker, it feels seamless. It feels like magic. This isn't something you can't automate all of these chemical profiles. We still have people doing some of this work. I think one day the AI will get there. Trust me. We'll definitely wanna use it. Right? It'll save us a a lot of resources there. But until that point, right, it's really important to us that this is a high quality, trustable data, not just, like, random data, not just, you know, pulled out. It's a trustworthy, right, validated source here, and I think that's gonna write power reports here. Yeah. And, you know, when you think about the reporting, so now you know everything about this chemical. You can actually start to automate where it belongs in your regulatory reporting, which reports does it belong on, what do we need to know about it, even things as simple as what volumes do we have across a single control area. All of those things become critical because those are the manual touch points. If that doesn't automate and someone misses that, you're obviously not putting your chemicals into the proper reports. And I think to summarize this is a lot of what Serena said. We look at manual touch points. Manual touch points in disparate systems create gaps, gaps create risk. One thing that we focus on all the time when we look at this from a product lens is how can we shine light on the areas that are a known gap or a known risk? Just shining light and knowing it's there doesn't mean we've actually fixed it. But now we can say, okay, now we know where the gaps are and where the issues are and where the problems are, and our team will get to work on figuring out how do we connect this together, how do we automate it so that you confidence in your compliance records and your reporting and you feel good that there is this seamless triangle without all that manual touch, which ends up creating the gaps. And to tie this all together, you know, we think of this trifecta. It's not just a trifecta of chemical inventory. It's actually a trifecta of the roles that it impacts. If the scientists don't want to use the tool at intake and they don't want to engage with it and it's not built for them and it's clunky or the UI is terrible, we hear that all the time. If someone says, hey, we're looking for a new solution. The scientists just can't use this. It's clunky. It's old. It's not working. They're not gonna use it. That's the reality. That has a a, you know, downstream impact on lab operations. They don't know what inventory is about to run out. Then they come back and say, lab ops, where's my chemicals? Lab managers, where's my chemicals? And they're not able to do their job effectively. That in turn, EHS doesn't know what's in the lab. It's not going into your regulatory compliance. And then somebody orders the wrong thing, And now you're over your MAQs and you're out of your fire code compliance, and it's not showing up on reports. So think about this in terms of you you wanna audit your program to figure out, do we have these three core components in place? However we get there, even if you take a step into it and it's not fully automated, get the three core components in place, then look at your roles throughout the lab and think about scientists, lab operations, and EHS and the impact it's having on them. Alright. So we know, right, this matters for everyone. We need the science to keep going. Right? Let's if you can't find what you're looking for, can't order what you want, running into issues here. Yeah. So, you know, thinking about this sort of, trifecta here of safety and compliance, we've actually been running a benchmark study across now thirty two. And I think this morning we actually had our thirty third one come through. So this is an ongoing benchmark we've been looking at. And the key here is this actually helps us from a product standpoint. How do we continue to drive down the time it takes someone to perform a task? How do we continue to make things more efficient? But also ensure that the data you know, when a customer looks at where they were day one, at the starting point before they turned on SciSure, you can do this with whatever solution you have, take an audit of your program. But what we did was we said, where are our customers day one and where are they at six months, one year, eighteen months? Not only how much time do we save them, but let's look at their compliance rates. How many, training requirements are met because we're tracking things more clearly? How often are are reports generated that are accurate and on time? So we did a deep benchmark study across a number of our customers, which we'll embed in the data here throughout the conversation. Yeah. Data is key here. Right? You can't measure. You can't change it. So let's talk about getting data in. Right? How do we get inventory data in? First, ChemSnap AI. I talked about this very briefly, right, with just a photo. It's kind of the stuff of dreams. Right? You take a picture of the bottle, the data inputs in there. You can also use barcodes and RFID tags. This is gonna help with that reconciliation. Right? Because inventory, we wish it was a onetime process. It's not. Right? You're always gonna have to be updating that data. Let's look through it. How do we add inventory in? This is with ChemTracker here. Add with camera ChemSnap dot ai. It's our most popular way to add inventory today. It'll auto populate a number of these fields from that label. Right? Name of that chemical connects it to our database. We're gonna see here. We're gonna have predefined drop downs about keeping our data clean, about who's owning this, where it's stored. And if I I am using barcodes or RFID tags, ChemTracker out of the box, you can use either or a combination. I see some of that, data scanned in here as well. Okay. Let's talk about data. Right? I just said you can't measure it. You can't change it. We saw with our customers, right, almost a seventy four percent reduction in the amount of time to add a container. Might be thinking, oh, this is just a few minutes, but, you know, this efficiency matters. Right? It's it's through one container that'll scale across, right, hundreds, thousands, right, hundreds of thousands. We see inventories in the two, three hundred thousand dollar three hundred thousand bottle range. I guess a lot higher in the in the dollar numbers there. And then that's about just adding inventory and reconciliation. I just say this number blew me away. Right, John? I'm sure for you too. Right? We see twenty hours to half an hour. So, like, ninety seven and a half percent. Crazy. Yeah. It's awesome to see that. I think, you know, one part of this is, hey. The technology can do great things, but the team that that's working in the technology, as Serena said earlier, you know, we're empowering people to be much more efficient, effective in their job to drive safety, to drive compliance. As she mentioned, know, when we look at the data, we're really trying to figure out that compounding effect. What's cool about this is you have a really this snowball effect or compound interest of the effort you put in upfront. Think about how many times across your institution or organization someone is adding a container to inventory. These stats say six point five minutes to one point seven minutes. Yeah, it's cool if it's a few times, but what we're talking about here is the scientist who's saying, I got to go perform this task. And every single time I do it multiple times a day, a week, a month, a year, I'm gaining back five minutes or so every single time I do that. You multiply that by the number of scientists you have across your organization, and that's really where the compound interest lines up. It's that power of a program that you put in place. Because today, even with ten scientists, if you're giving them a simple solution to use, they will be very grateful for that five minutes. When you compound it and you start to scale and grow, you have exponential ROI to the organization. And reconciliation rate twenty hours. This is, more than two day two two business days. Right? So how do we how do we get these numbers? Right? Accuracy of automation. These are also better records we're getting. Right? It's easy to use. We're gonna have this hazard data in place. Right? So we're gonna see more time saving even once that inventory is in because when you add inventory, it'll connect to our validated chemical profiles here. So let's talk about these chemical profiles. Right? This is this is the key. When you enter data or when we import your data, right, from spreadsheet, we're gonna tie to our chemical property database. This is available only in ChemTracker. It's you know, we have about two million safety data sheets with auto matching to safety data sheets. We provide hazards, storage groups for chemical compatibility, density data. That's gonna be key for fire codes, CAS numbers, and synonyms. Right? So any name on that label, wanna make sure we can look it up. Right? One person enters dichloromethane, another person enters methylene chloride. Now you can search by either name and find that. We'll do unit conversions and then bring in all this regulatory data for you. So this, again, it all comes in from the chemicals. Right? Here we are at this example I was just talking about, we can type in even that abbreviation DCM. This is one, right, again, for researchers. This is also key for each one of these profiles. This is an example for dichloromethane. It's definitely one of our hot chemicals on the mind after that EPA rule. Right? Common solvent turned a high hazard, high abbreviation, high supervision here. We have structure data. You could also add SOPs with safety data sheets. Physical properties. Right? Densey mentioned this is key so that you can do those unit conversions automatically. Storage handling for EPA tier two reports. And this is then just regulations just for this chemical. Alright? This is state, local, DOT. I won't say we have every single regulation in the United States ever, but we have a lot of them. Right? There's a lot here. And, again, these are the ones for just one chemical. Highlighting here right now is the ones for fire code reporting. We know those. Right? Not always one to one with GHS. We have that available for our customers in the database. And all of this comes together, right, to build in, these validated profiles. Hey, Serena. I did wanna pause for a second. We have a a question in the chat that I think is worth clarifying. Someone asked, hey. If I if I use the photo and I take a photo of the chemical container, will it actually bring the chemical profile over to the container and assign the SDS to it? You're saying I can do that with a photo. That's the beauty of the trifecta of what we built here. You know, just to tell a story to really drive this home, this idea came about. We were at a site visit with someone who was about to deploy, ChemTracker. They had not yet turned it on. And while we were walking through on the prep meeting, we had a scientist take a box of chemicals over to the bench and start pulling them out and hitting their keyboard. The head of safety said, let's go take a look at what's going on here. Turned out she was searching the chemical profile like, hey, what hazards am I getting exposed to here? Walking through a list and jotting it down on a note on her desktop so she knew what she was working with. And, you know, the Chemsnap AI AI idea came out of those conversations where we said we have to do better. We cannot have our scientists spending any time on this. We want them to come in, take a picture of the container, hit the database, grab the SDS, tell them everything they need to know, and then one step further, put that into the health and safety and regulatory compliance reports. So something as simple as once that's in there, I just wanna search for my peroxide formers. You know? That's the beauty of this feature. So that's called ChemSnap AI, and that's something that's available now. Basically kicks off everything that we see here. Yeah. So this this is what makes it scalable. Right? You know, we've seen inventory everywhere. Right? Your lab is not a store. It's not generic inventory. You need to understand those chemicals, and that is what ChemTracker does here, whether that's with photo, whether that's with spreadsheet, whether, you know, maybe you're typing a catalog number if you you don't have a mobile device here. So we have all that together right here. Alright. If we're running again, right, how do we get these numbers? It's from, right, again, the the multiple steps here, that trifecta. So we have ChemSnap. We have other ways to get the data and bar codes and RFID options to make reconciliation easier, tie to those validated chemical profiles with SDS auto match, and then you can pull these, you know, these real time reports and queries. Again, correcting chemical data, looking at over two work days a month. Right? Think about what else you can do with that. Right? Do with that time here. So when we think about real time reporting, right, this is the end game. This is what we're trying to get to. Ninety nine point four percent time savings that we've seen, and this is, again, with a real customer study here going from over twenty four hours to seven point two minutes to to pull report. This stat actually didn't surprise me when I'd seen this, right, collected in. Think about, you know, just even someone asking, okay. You know, what are all our peroxide formers? What do we have that's, you know, like a certain level of toxicity and then doing those unit conversions? I've seen firsthand for customers, right, out how hard this is if they don't, you know, I don't have a program that can do this like ChemTracker does. We can do tier two. We can do RTK, including threshold calculations. For our code report, you'll have your control areas. You can see percentages of MAQs here. Right? Because we have this data in here, and you can do it by whatever location set you need to here. In terms of that real, you know, real time reporting here, I have over nine hundred regulations, these automated hazard calculations with the MAQ ones. Right? Again, fire code is is often everyone's brain, especially in the Boston area here. A lot of ways you can get to this type of data, whether it's, you know, export to spreadsheet or API. And what we're showing here, this is exactly what it would be like for you. Right? Once we have your data into ChemTracker, if you're not a customer already, select any regulation in our database and convert into exact units you need. Labs have everything in metric units. You can run-in pounds, gallons, and cubic feet here and get the data that you need out. Yeah. So, Serena, we can go to the next slide. This is a refresh on the on the trifecta and the triangle here. We're just gonna walk through a little bit of, you know, in the detail of what we covered as a recap. But when you think about ChemTracker, you know, we built it for this trifecta. The whole point is that if we can't create this seamless experience from the efficient intake to that validated chemical profile that you can trust. You know, this is an area we spend a lot of time and a lot of resources at SciSure investing in that chemical profile database, so that our customers don't have to think about it. It's an out of sight, out of mind. You license SciSure and ChemTracker, not something you have to think about. And then moving into real time reporting, did see there's so much in the chat. A question about, we report by laboratory? Can we report by building? Absolutely. You'll start to see the data broken out in different groups so that you can query it. You can find out, search the data. I want to look for pyrophoric, so I want my peroxide formers. I want to know where they are. I want to know who's working with them. All those things that you can query the data. And on top of that, you can push all of it into your regulatory reporting. So we talked earlier about our goal is to get rid of those blind spots and those gaps. You have to first start by shining a light on what it is, and that's probably the exercise I'm sure most of you will go back and do. Hey. What do we have today, and how do we get ourselves to a point that we can meet this sort of trifecta, process here? And, really, it's not just efficiency and speed. It's the accuracy and the productivity that we're after. This is what we do. We understand chemicals. So I think Mark's gonna be joining here to help us with the q and a. John, maybe you can give a little overview of this while we're pulling up our questions here. Yeah. So if you think about, we just wanna you know, many of you come to us from different parts, whether you're an existing customer or future customer. We have our research side of the house, which is your ELN, LIMS, sample management, all the solutions and tools to really help the scientists and researchers, advance the science, innovate, and do it safely and efficiently and have reproducibility in their daily work. And then we have the health and safety and lab operations side. We have a number of solutions. We also have marketplace and integrations. I think I saw some vendors joined us today and partners. If you'd like to integrate your solution into SciShore, we have an open marketplace and ecosystem where vendors can plug in. That gives our customers access to a number of different solutions to continue to build that seamless experience inside of SciShore. Yeah, Mark, if you don't mind me jumping in, saw this question from Jackie here that I really wanna start with. Jackie wrote, I also wonder since the chemical hazards are classified from using GHS from the SDS while the MAQ report follows IFC, does that mean there's a link mapping between GHS and IFC in the system? So for anyone not familiar, IFC is international fire code, basis of most, you know, most fire codes in the state states and cities in the United States. So our chemical database is different than others in that it's not data just blindly pulled from safety data sheets. I wish it was. Right? That'd be a lot easier to do. Fire code data in particular, most of the time, right, it's not one to one with the GHS classifications. This is for things like toxicity in particular. We have this is, I'd say, the hardest thing to train for our curators for the database. They're looking at LD 50s, LC 50s, calculating it into the right time range, things like that to get those fire code assignments, right? Because unfortunately they're not on the safety data sheets here. That's great. And also guys, there's another question. It's regarding MAQ again from Erica. She's mentioning for MAQ calculations, does this software allow for assigning laboratories a percentage of the MAQ threshold? Yeah. There's a way you could set this up. You actually have control over what the MAQs are for a given control area or control zone. So in that case, probably the way that we'd set up the location so the labs could be limited just a certain section of the room and basically have their own their own MAQ percentage there. Mark, I wanna grab a real quick one here. I see that. Yep. Contractors working on-site utilize ChemTracker, or can we give them access IDs and passcodes? Absolutely. We work with almost any vendor that you have on-site now, we work with. We always say we play nice in the sandbox. If their job is to get your program compliant to be safe, to be efficient, we are the tech stack that helps. So actually many of the sites you might, go to or visit, it's often our software powering the effort going on there. And then we work with contractors, vendors, consultants to make sure that they have best in class technology to do their job. K. And I see a few a few questions about SDS updates. I see one from Mindy asking if it will notify stakeholders and chemical owners if there's an SDS update. One from Jackie about the SDS and ChemTrackers database updates automatically when a new version is released. So what we have every right? We know SDS. Right? This isn't a onetime job. So each each chemical, each bottle, right, we recheck to see if there's a newer SDS or a better match at least once a year. They'll automatically update that within the system. Not notification of that per se, but you'll be to see that, right, see that SDS and see the last date that that was was updated there. Someone also asked, I wanna answer this one about reconciliation. I would ask this. That's a great system. Thank you, I would. It's really kind. Said after I have my inventory done, can I multiscan the bottles for reconciliation? There's something you can do with barcodes or RFID tags. You know? Right? You might have, like, ten thousand bottles in one lab. So it's designed to designed to scale there. And I also see another important question that you might be able to touch base on, Sarina. The question is, can you import my data if I'm currently using another system? Yes. Absolutely. Say, it's pretty rare that, you know, a new customer comes in and they don't have anything for inventory on occasion, but usually folks at least have a spreadsheet. Most software systems out there can provide some sort of export to spreadsheet, and we can work with you to figure out, right, how that fits in our template. We don't have quite as many required fields as some systems do to, you know, to import. So typically, right, even if you don't have CAS numbers, we can import your data, and we can match to the database so you can get those safety data sheets. You can get that hazard data and those reports right away here. Yeah. Sarina, I just wanna go add to that answer. So one thing that's really important is during your discussion with our team, we'll do really a discovery call and work with you to diagnose what problems are you facing, what's existing today, how do we improve it, how do we set up metrics so that we can track the output, not just stand up the system, but then as we shared earlier, monitor our progress. One thing that we will do is we always assign a dedicated implementation team to every single customer. So that team will help you with getting any data into the system, setting it up the right way, checking back with you, making sure that you have this highly successful implementation and deployment. And in the past ten years, we have one hundred percent success rate in deployment and industry best retention. That's not a coincidence. It's because of the effort we put in to understand what you need from us to build a program that meets those needs and then to come back and implement it and deliver it the way that you wanted us to deliver it to your success criteria. So if there's any questions around that, I'm happy to set up a call. We do not want to lose customers, and we take that to heart. Some companies will say, Hey, we're going get ten through the door, we'll lose a couple as we go. We do not want to lose a single customer. So we put a ton of effort into that implementation, making sure we get it right. If you get it right out of the gate and you're successful, it works in perpetuity. If you don't get it right out of the gate and it's a tough rollout, it's not going to work. And that's why our model is to be very white gloved and hand holding out of the gate to make sure that we get to success quickly. I see another question here I want to touch on. Rui Feng, I apologize if I don't have the pronunciation of your name right here, but could you please introduce the function of tracking time sensitive chemicals if you have? Yes. So among the lists we have in ChemTracker, we have one that's time sensitive that might cover things, you know, that with, you know, with age could degrade in a way that could be hazardous, as well as a separate list of peroxide formers. So absolutely something you can get to here as well as tracking expiration dates, seeing what's expired, all of that here. Right. Yeah. I mean, we still have a full house here. We still have a lot of questions, but I think in in the interest of time, Mark, maybe we'll wrap up here. We'll coordinate, some follow-up on the remaining questions here. Thank you so much everyone taking the time Thank you all. Really appreciate it. Have a great day.

Who needs Safety Data Sheets and when are they required?
Under OSHA's Hazard Communication Standard, SDS requirements apply across the chemical supply chain:
Chemical manufacturers and importers must prepare an SDS for every hazardous chemical they produce or bring into the country and provide it to downstream customers with the first shipment.
Distributors must ensure the correct, current SDS accompanies every shipment of hazardous chemicals to downstream buyers.
Employers must obtain and maintain a current SDS for every hazardous chemical present in the workplace. These must be readily accessible to employees during every work shift, not stored in a locked cabinet or buried in a shared drive folder that nobody can find during an emergency.
This requirement applies broadly. It covers research laboratories, manufacturing facilities, hospitals, agricultural operations, maintenance shops, and any workplace where employees handle chemicals beyond typical consumer use. OSHA citations for Hazard Communication violations are consistently among the agency's most frequently issued, and penalties for serious violations can reach $16,550 per instance.
For scientific organizations managing hundreds or thousands of chemical containers across multiple labs, buildings, or campuses, maintaining current SDS documentation is a significant operational challenge that manual processes alone cannot reliably support.
How to manage Safety Data Sheets effectively across your organization
Having SDS documents on file is only part of the requirement. The real compliance challenge is ensuring they are current, accessible, and connected to your chemical inventory. Here is what an effective SDS management process looks like in practice.
The difference between paper-based and digital SDS management isn't just convenience. It affects compliance readiness, emergency response time, and whether your team can actually find the right information when it matters.
- Keep SDS documents linked to your chemical inventory. An SDS is only useful when it can be quickly located for the specific chemical in question. The most reliable approach is an electronic system that automatically links SDS records to individual chemical containers, searchable by chemical name, CAS number, or manufacturer. This eliminates the common problem of having an SDS binder that doesn't match what's actually on the shelf.
- Review and update on a defined cycle. The GHS recommends reviewing SDS information every 3 to 5 years, even when no new hazard data has been reported. EU and OSHA regulations also require updates within 90 days of learning significant new information about a chemical's hazards. Without a systematic review process, outdated sheets accumulate and create silent compliance gaps.
- Ensure accessibility meets regulatory expectations. OSHA requires SDS to be readily accessible during every work shift. "Readily accessible" means employees can locate the relevant SDS quickly, without needing to track down a supervisor, search through folders, or navigate a confusing directory. Electronic SDS databases, particularly those integrated with chemical inventory management, meet this standard by allowing search-based access from any workstation, lab terminal, or connected device.
- Connect SDS to training and onboarding. Every employee who works with or near hazardous chemicals must be trained on how to read, understand, and access Safety Data Sheets. This training should be documented and refreshed whenever new chemicals are introduced or when SDS content changes due to reclassification. A training management system that tracks completion and triggers reminders simplifies this for organizations with large or distributed teams.
- Prepare for audits proactively. Regulators expect to see an up-to-date chemical inventory with matching SDS for every hazardous chemical on-site. They also check that employees know where to find SDS documents and how to read them. Organizations that manage SDS digitally, with integrated compliance reporting, can demonstrate audit readiness in minutes rather than scrambling to assemble paper trails.
For a deeper look at what regulators typically ask during chemical safety inspections, see 5 Common Questions From Chemical Regulators (and How to Address Them).
Organizations building or upgrading their chemical inventory program can also explore SciSure's Chemical Inventory Management Playbook for a step-by-step framework covering intake, reconciliation, SDS management, and regulatory reporting.
GHS hazard pictograms: visual communication of chemical risks

The GHS uses a set of nine standardized pictograms, each enclosed in a red diamond-shaped border, to visually communicate specific categories of chemical hazards. These pictograms appear on both chemical labels and in Section 2 of every SDS.
Each pictogram represents a distinct hazard type: health hazards (such as carcinogenicity or respiratory sensitization), physical hazards (such as flammability or explosiveness), and environmental hazards (such as aquatic toxicity). The visual standardization means that a worker in any country can recognize the hazard category at a glance, regardless of language.
For lab managers and safety and compliance officers, understanding pictograms is essential for proper chemical storage (ensuring incompatible materials are separated), for selecting appropriate PPE, and for making informed decisions about which chemicals can be used in shared spaces.
A reference guide to all GHS pictograms is available from OSHA's Quick Card on Pictograms.
Conclusion
Safety Data Sheets are one of the most important documents in any chemical safety program. They standardize how hazard information is communicated, protect the people who work with chemicals every day, and form the basis of regulatory compliance under OSHA's Hazard Communication Standard.
With OSHA's revised HCS aligning U.S. standards to GHS Revision 7 and compliance deadlines approaching in 2026 and 2027, now is the time for organizations to review their SDS management processes. That means verifying that every chemical on-site has a current SDS, ensuring employees can access and understand those documents, and connecting SDS records to the broader chemical inventory and safety infrastructure that supports day-to-day operations.
The organizations that treat SDS management as an ongoing operational process, rather than a one-time filing task, are the ones that stay audit-ready, respond faster in emergencies, and build a genuine culture of chemical safety.

About the author:
Jon ZibellJon Zibell is Vice President of Global Alliances & Marketing at SciSure, where he leads strategic partnerships with organizations like The Engine (MIT), US Lab Partners, and My Green Lab to help life science and research institutions modernize lab operations. He writes about the operational, safety, and technology challenges facing modern scientific organizations. Jon holds a B.S. in Marketing & Corporate Communications from Bentley University.
See all posts from this authorRead more of our blogs about modern lab management
Discover the latest in lab operations, from sample management to AI innovations, designed to enhance efficiency and drive scientific breakthroughs.
Sign up for our newsletter
Please check your email to verify your submission.



