LIMS Integration: How It Works and Key Benefits
Learn how LIMS integration works, key architectures, and operational benefits for regulated R&D labs, from implementation planning to audit readiness.

Download Whitepaper
TL;DR
- LIMS integration connects lab systems so data moves in a structured, traceable way
- Without it, labs rely on manual entry, leading to errors and audit gaps
- Proper integration aligns workflows, data models, and access controls for compliance
- It reduces admin work, improves data integrity, and speeds up lab operations
- Modern labs move toward unified platforms where ELN, LIMS designed for the R&D sector, and EHS share one data model
Ready to see SciSure in action?
No commitment · Free consultation
Regulated R&D laboratories rely on multiple digital systems, including the Electronic Laboratory Notebook (ELN), LIMS, instruments, ERP platforms, and compliance tools. The challenge is not whether software exists, but whether these systems exchange structured data in a controlled and governed way.
When integration is limited, laboratories depend on:
- Spreadsheets;
- Manual transcription;
- Fragmented audit trails.
When properly implemented, integration supports automated data flow, version control, and audit readiness, forming the foundation for reproducible and compliant R&D operations.
What is LIMS integration?
LIMS integration is the traceable exchange of structured data between a LIMS and other laboratory or enterprise systems. It ensures that sample records, instrument outputs, inventory data, and compliance documentation remain aligned without manual reconciliation.
In regulated R&D environments, this exchange must follow controlled schemas, audit logging, and role-based access to maintain data integrity and compliance.
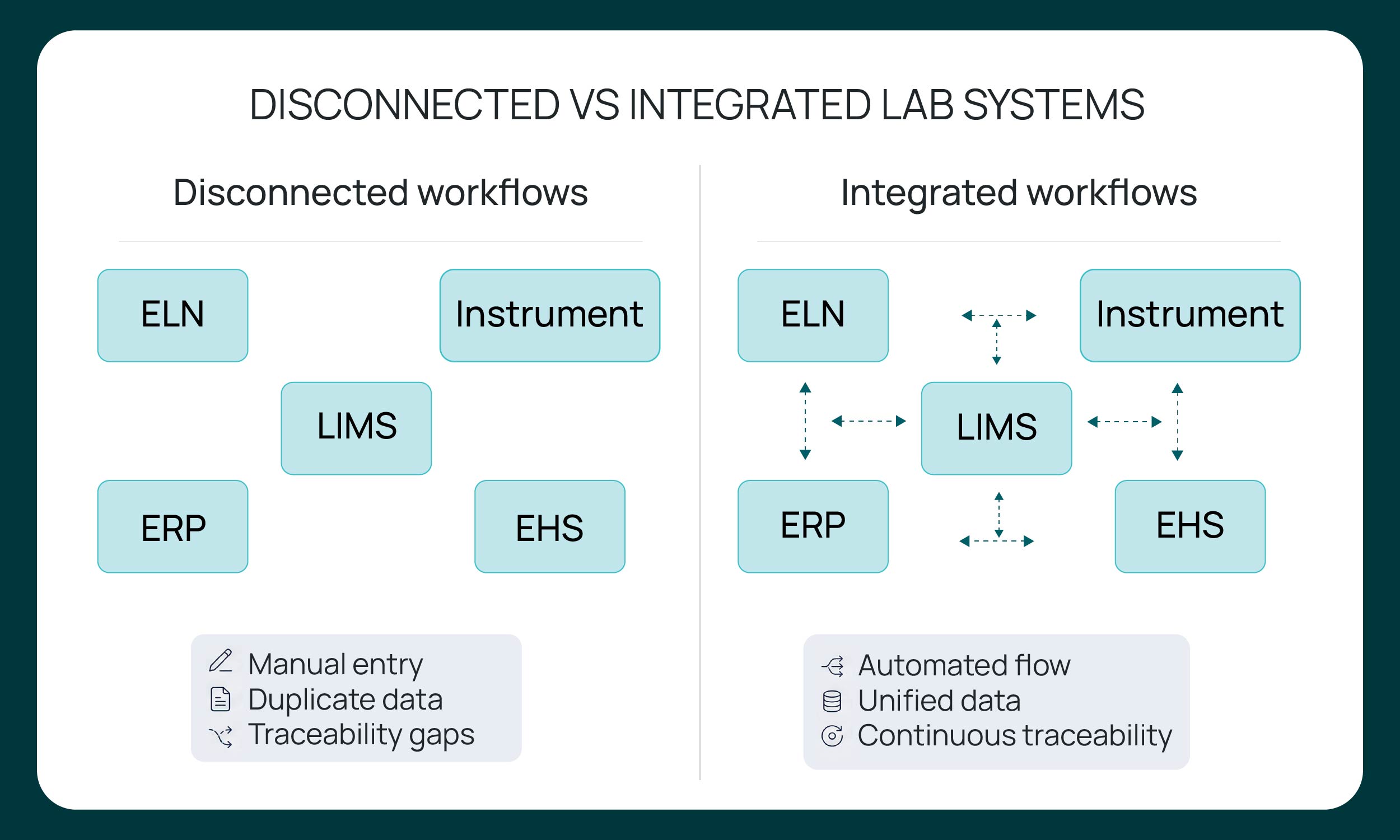
What LIMS integration connects across Laboratory Infrastructure
LIMS integration operates across multiple layers of laboratory infrastructure. It connects systems that manage experimental design, structured sample data, instrument outputs, inventory records, enterprise processes, and regulatory documentation.
At the operational level, integration connects the full sample lifecycle. Sample registration, metadata, status changes, storage location, and lineage must remain consistent between the LIMS and adjacent systems like the electronic lab notebook, instrument platforms, and inventory tools. Without integration, these records diverge, creating reconciliation work and traceability gaps.
Instrument integration ensures that analytical results, calibration data, and test outputs flow directly into the LIMS under defined schemas. Enterprise integrations connect laboratory data to ERP, procurement, and financial systems, enabling synchronized batch tracking, purchasing, and inventory thresholds. Compliance-related integrations align audit logs, training records, and regulatory documentation with laboratory activity.
It is important to distinguish between structured operational data and unstructured experimental context. Structured data includes sample IDs, timestamps, method parameters, and status fields that must follow controlled schemas. Unstructured data includes scientific narrative, observations, and exploratory notes. Effective LIMS integration governs the structured layer while maintaining associations to contextual information, preserving both traceability and scientific integrity across systems.
How LIMS integration works step-by-step
LIMS integration is not a single configuration task. It is a structured component of LIMS implementation that requires planning, validation, and governance. In regulated R&D environments, integration must align with documented workflows and compliance requirements.
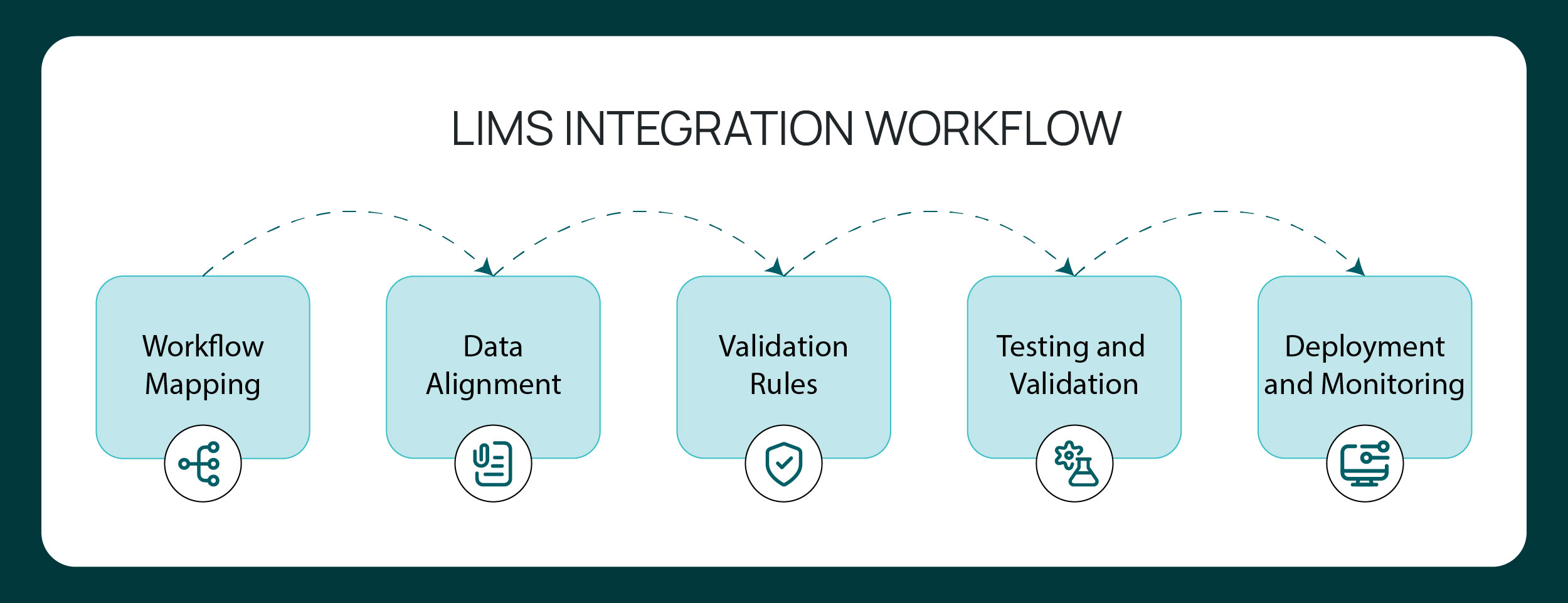
A typical integration sequence includes:
- Workflow mapping: Laboratory processes are mapped end-to-end to identify where data originates, how it moves, and which systems require synchronization. This includes sample registration, instrument output capture, inventory updates, and reporting.
- Data model alignment: Data fields, identifiers, and metadata definitions are aligned between systems. Sample IDs, method parameters, timestamps, and user roles must follow consistent schemas to prevent ambiguity.
- Metadata and validation rules definition: Structured fields are defined with validation logic to ensure data integrity. Required fields, controlled vocabularies, and formatting standards are documented prior to deployment.
- Role and permission mapping: User access levels and authentication controls are configured to align with regulatory expectations. Role-based access and audit logging must remain consistent across integrated systems.
- Testing and validation: Integration workflows are tested under defined scenarios. In regulated environments, validation documentation supports 21 CFR Part 11 and GxP requirements, ensuring traceability and reproducibility.
- Deployment and monitoring: Once validated, integrations are deployed with change control documentation. Ongoing monitoring ensures continued data consistency and system performance.
An effective LIMS system implementation treats integration as infrastructure, not as an afterthought. Proper documentation and validation during LIMS implementation planning reduce risk and support long-term audit readiness.
Why LIMS integration is no longer optional in regulated R&D
In regulated environments such as biotechnology and pharmaceutical R&D, disconnected systems introduce operational and compliance exposure. Laboratories operating under regulations such as 21 CFR Part 11 must demonstrate data integrity, traceability, and controlled audit trails. Fragmented infrastructure makes this difficult to sustain.
When LIMS integration is limited or absent, common workflow risks emerge:
- Errors in instrument data entered into the LIMS
- Duplicate data entry between the electronic lab notebook and sample records
- Inconsistent metadata across systems
- Gaps in audit log continuity
- Delays in retrieving documentation during inspections
These issues are not technical inconveniences. They affect data reliability, review cycles, and regulatory defensibility.
Regulatory frameworks do not mandate specific software architectures, but they require:
- Complete and attributable records
- Controlled version histories
- Secure user access and role management
- Traceable data modifications
- Reproducible workflows
Without integration, laboratories rely on manual reconciliation to meet these expectations. With structured LIMS system implementation and governed integration within modern LIMS software, compliance controls become embedded within daily workflows rather than applied retrospectively.
In modern regulated R&D, LIMS integration is part of the laboratory’s operational infrastructure, directly influencing reproducibility, audit readiness, and long-term scalability.
Types of LIMS integration architectures
LIMS integration can be implemented through multiple architectural approaches, depending on system maturity, regulatory requirements, and scalability needs. The integration model selected directly affects governance, maintainability, and long-term performance.
API-based integration provides structured and scalable system connectivity.
API-based integration uses REST or similar interfaces to exchange structured data between systems in a controlled and versioned manner. APIs define schemas, authentication protocols, and validation rules that support secure data transmission. Proper documentation and schema mapping are critical to ensure long-term governance, especially in regulated environments where auditability and change control must be maintained.
Direct database integration offers flexibility but requires strict controls.
Direct database integration allows systems to exchange data at the SQL level, often enabling rapid implementation. However, this approach can introduce governance risks if access controls, validation logic, and change management processes are not strictly enforced. Without clear data ownership and documented schema alignment, database-level integrations may compromise data integrity and security posture.
File-based exchange supports simple integrations in controlled environments.
File-based methods use structured formats such as CSV, XML, or JSON to transfer data between systems through scheduled imports, exports, or monitored directories. This approach is effective for stable, well-defined data flows in controlled environments. However, it becomes limited in dynamic or high-throughput laboratories where real-time synchronization and validation are required.
Middleware platforms normalize communication between multiple systems and instruments.
Middleware functions as a translation layer between instruments, enterprise systems, and the LIMS. It standardizes communication protocols, transforms data formats, and routes information according to predefined logic before transmission into the core system. Middleware is particularly useful in complex laboratory environments with diverse instruments and legacy systems requiring harmonized integration.
Core integration points in modern laboratories
Modern laboratories rely on multiple systems to manage research execution, operational logistics, and compliance oversight. Effective LIMS integration aligns these systems through structured data exchange across defined integration points.
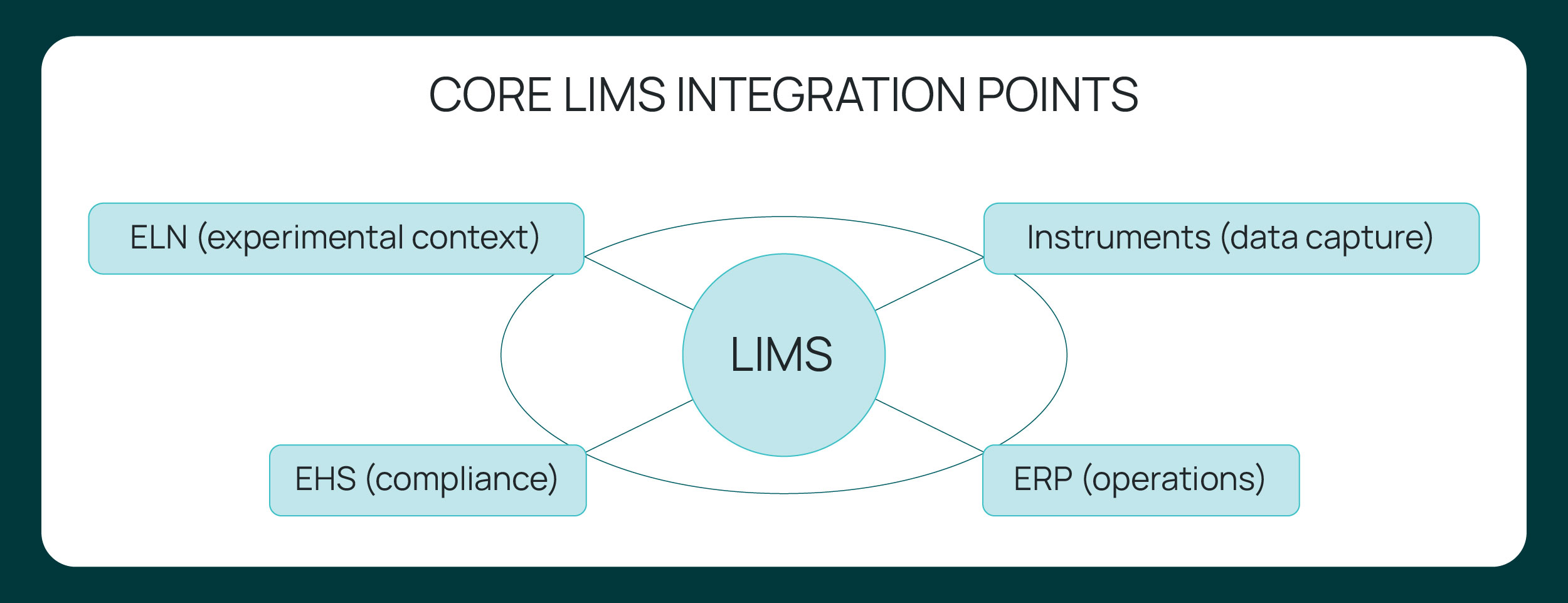
ELN and LIMS integration connects experimental context with structured sample data.
The electronic lab notebook captures experimental design, scientific narrative, and contextual observations, while the LIMS manages structured sample identifiers, workflows, status tracking, and inventory records. Integration links experiment records directly to sample lineage, ensuring that protocols, results, and material movements remain associated under version control. This structured association improves traceability, reduces reconciliation work, and strengthens reproducibility across studies and teams.
Instrument integration enables automated data capture and eliminates transcription risk.
Analytical instruments such as balances, LCMS platforms, PCR systems, and other devices generate structured outputs that must be recorded accurately within the LIMS. Through APIs, middleware, or controlled file parsing, instrument integration transmits results directly into predefined data fields. Automated metadata capture standardizes timestamps, method parameters, and operator attribution, reducing manual intervention and improving data consistency.
ERP and procurement integration aligns laboratory operations with enterprise systems.
Laboratories operate within broader financial and supply chain frameworks. Integration between the LIMS and ERP systems synchronizes purchasing data, batch release status, and inventory thresholds without duplicate entry. This alignment provides operational visibility into material usage and spending, supports procurement planning, and ensures that laboratory activity is accurate within enterprise reporting environments.
EHS and compliance integration is emerging as a way to embed regulatory oversight into daily workflows.
Chemical inventory records, SDS documentation, inspections, and training activities often exist in separate compliance systems. As laboratories evolve, integration is increasingly linking these elements to sample and experiment data within laboratory workflows. This approach keeps safety classifications, inspection status, and regulatory documentation aligned with operational activity, moving compliance closer to continuous and structured oversight rather than reactive documentation.
LIMS implementation planning for integration success
Successful LIMS integration begins before any technical configuration. LIMS implementation planning must define governance structures, architectural standards, and cross-functional ownership to ensure long-term stability.
Integration planning typically involves coordination between:
- Lab Operations
- IT and security teams
- Compliance and quality stakeholders
- Research leadership
Early alignment clarifies data ownership, system boundaries, and validation expectations.
Key planning components include:
- Architecture review: Evaluate existing systems, instrument interfaces, ERP connections, and compliance tools to determine integration scope and technical constraints.
- Risk assessment: Identify regulatory exposure, data integrity risks, and operational bottlenecks that integration must address.
- Data migration and normalization scope: Define which historical records require migration, how metadata will be standardized, and how legacy inconsistencies will be resolved.
- Validation and documentation requirements: Establish documentation protocols for configuration, testing, and change control in alignment with GxP and 21 CFR Part 11 expectations.
- Change management and training: Prepare users for workflow adjustments, new system interactions, and updated documentation practices.
Without structured planning, integration efforts often become reactive and fragmented. When governed properly, LIMS implementation planning ensures that integration strengthens reproducibility, audit readiness, and operational continuity rather than introducing additional complexity.
Common failure points in LIMS integration
LIMS integration challenges rarely originate from technical capability alone. In most cases, failures result from gaps in planning, governance, or cross-functional alignment. When integration is approached as a series of isolated technical tasks rather than as infrastructure design, long-term instability often follows.
Over-customization is a frequent issue. Extensive modifications to data models or workflows may address immediate operational needs but complicate validation, upgrade cycles, and documentation control. Similarly, legacy systems that lack standardized interfaces can introduce hidden constraints if not identified during early LIMS implementation planning.
Unclear data ownership also creates integration risk. When responsibility for schema governance, metadata standards, and change control is not formally assigned, inconsistencies emerge across connected systems. In regulated environments, inadequate documentation and insufficient user training further undermine audit readiness, as users may revert to manual workarounds that bypass structured workflows.
Sustainable LIMS integration depends less on technical complexity and more on disciplined planning, defined accountability, and documented validation procedures.
LIMS integration benefits in operational terms
Well-designed LIMS integration produces measurable operational improvements. These benefits are not abstract efficiencies; they directly affect documentation burden, audit readiness, and data integrity across regulated R&D workflows.
Reduced manual entry
Automated data exchange between instruments, the electronic lab notebook, inventory systems, and the LIMS minimizes duplicate entry and transcription. This reduces administrative workload and lowers the probability of human error in regulated records.
Stronger audit trails
Integrated systems maintain continuous, synchronized audit logs across workflows. Sample changes, result updates, user actions, and status transitions remain attributable and time-stamped, improving inspection readiness and documentation defensibility.
Improved turnaround time
When data moves automatically between systems, laboratories spend less time reconciling discrepancies. Review cycles accelerate because sample status, instrument outputs, and compliance documentation remain aligned in real time.
Continuous compliance
Integration embeds validation logic, metadata controls, and role-based permissions within daily workflows. Compliance shifts from periodic preparation to structured oversight supported by consistent data governance.
Enhanced reproducibility
Structured associations between experiments, samples, inventory, and regulatory records improve traceability. Version-controlled data and synchronized metadata strengthen reproducibility across teams, studies, and sites.
In operational terms, LIMS integration benefits translate into reduced administrative burden, improved audit readiness, and sustained data integrity within regulated laboratory environments.
From integration to unified infrastructure
Point-to-point LIMS integration connects individual systems. However, stitching together tools through multiple interfaces can still result in fragmented oversight if each system maintains separate data models, audit logs, and governance controls.
A more unified architectural model is beginning to emerge. This model reflects the direction of a Scientific Management Platform (SMP), where ELN, LIMS, and EHS workflows are brought closer together within a shared environment.
Rather than relying only on synchronization between disconnected platforms, laboratories are moving toward connected interfaces where users can operate across research, inventory, and safety workflows within a consistent system environment. This reduces friction between systems and improves visibility across operational and compliance activities.
This distinction is structural. In stitched environments, integration maintains alignment between systems that remain independently governed. In more unified models, platforms increasingly standardize access, interfaces, and selected workflows, reducing the reconciliation required between systems.
In practice, this shift often begins with specific domains. For example, shared modules such as equipment management or inventory workflows can be unified first, allowing data and processes to align incrementally across systems.
Over time, this approach supports closer alignment between experiments, samples, inventory, and compliance records, even when underlying systems remain partially independent.
For regulated R&D organizations, this architectural direction supports reproducibility, audit continuity, and scalable infrastructure, while allowing systems to evolve without introducing unnecessary complexity.
Evaluating your current LIMS integration strategy
Laboratories often assume integration is sufficient because systems exchange data. A more accurate evaluation focuses on governance, traceability, and scalability.
Use the following questions to assess integration maturity:
- Is integration formally documented? Are workflows mapped, version-controlled, and validated under change management procedures?
- Are audit logs continuous across systems? Do user actions, data modifications, and sample status changes remain attributable when data moves between platforms?
- Is instrument integration validated and standardized? Are metadata schemas defined and enforced, or do manual file transfers introduce inconsistency?
- Is workflow traceability end-to-end? Can a sample be tracked from registration through experiment execution, inventory movement, and compliance documentation without manual reconciliation?
- Are roles and permissions aligned across systems? Do access controls remain consistent between connected platforms?
- Is the architecture scalable? Can new instruments, sites, or regulatory requirements be added without redesigning integration logic?
If the answer to several of these questions is unclear or undocumented, the issue may not be connectivity, but governance maturity.
Effective LIMS integration supports structured oversight rather than isolated system alignment.
How SciSure supports LIMS integration
SciSure supports LIMS integration as part of a Scientific Management Platform that connects the electronic laboratory notebook; LIMS; and Environmental, Health, and Safety (EHS) workflows within a shared data model.
Instead of relying on point-to-point integrations, structured data across experiments, samples, inventory, and compliance records remains aligned by design. This reduces manual reconciliation and improves traceability across the full sample lifecycle.
Through APIs, integrations, and marketplace add-ons, laboratories can connect instruments and enterprise systems while maintaining controlled schemas, audit logging, and role-based access.
Because integration is built into the platform architecture, laboratories maintain data integrity, audit readiness, and reproducibility without adding additional system complexity.
LIMS Integration as Lab Infrastructure
LIMS integration determines whether a laboratory operates with fragmented systems or with structured, traceable workflows. In regulated R&D environments, integration directly impacts reproducibility, audit continuity, and data integrity.
When implemented with governance and validation, integration aligns sample lifecycle data, instrument outputs, inventory records, and compliance documentation under controlled schemas and role-based access. It becomes part of the laboratory’s core infrastructure rather than a technical add-on.
For modern R&D organizations, structured integration provides the connected foundation required for scalable and compliant scientific operations.
SciSure reflects this approach by unifying the electronic laboratory notebook, LIMS designed for the R&D sector, and compliance workflows within a shared data model, allowing integration to function as part of the underlying infrastructure rather than a separate layer.
FAQ
Is a LIMS the same as an ERP system?
No, a Laboratory Information Management System (LIMS) is not an ERP system. A LIMS is designed specifically for laboratory environments, where it manages sample tracking, workflows, and structured scientific data. In contrast, ERP systems handle broader business processes such as finance, procurement, and supply chain management. While they serve different purposes, the two are often connected through integration, allowing laboratory data to align with enterprise-level operations.
What are the benefits of integrating lab instruments with a LIMS?
Integrating instruments with a LIMS improves both accuracy and efficiency by allowing data to flow directly into structured records without manual entry. This reduces transcription errors and ensures consistency across workflows. It also speeds up turnaround time, since results are immediately available for review and reporting. In regulated environments, instrument integration strengthens audit trails by automatically capturing metadata such as timestamps, methods, and user actions.
Why do modern labs need a LIMS?
LIMS are important because they bring structure and control to complex laboratory workflows. They help ensure data is consistent, traceable, and audit-ready, while reducing the reliance on manual processes.
How to choose the right LIMS?
Choose a LIMS based on how well it fits your workflows, integration needs, and regulatory requirements. In regulated environments, a LIMS designed for the R&D sector with strong data governance and scalability is essential.
Read more of our blogs about modern lab management
Discover the latest in lab operations, from sample management to AI innovations, designed to enhance efficiency and drive scientific breakthroughs.




